v/acc: PopVax’s plan to save 1 million lives each year by massively accelerating vaccine development
The million lives mission
Vaccines are among the most successful public health interventions known to man. They’ve saved millions of lives each year over the past century, with an estimated total of 150+ million lives saved by childhood vaccines from 1974-2024, and further wide deployment of recently-developed vaccines such as RTS,S and R21-Matrix M against malaria is on track to save millions more in this decade alone.
When a pandemic pathogen such as SARS-CoV-2, which causes COVID-19, emerges, vaccines are a key tool in ensuring, as Vitalik Buterin wrote in his d/acc – defensive accelerationism – manifesto, that societies don’t have to “resort to the sledgehammer of social compulsion,” including deeply restrictive measures such as lockdowns that circumscribe freedom to a degree that I, and most other people across the world, find unacceptable. The COVID-19 vaccines are estimated to have saved as many as 20 million lives during the acute phase of the pandemic, and if a broadly-protective betacoronavirus vaccine had been available prior to 2020, something that was likely technically feasible by leveraging work done in the aftermath of the original SARS epidemic, then perhaps the deaths and damaging disruptions of our recent pandemic era would never have happened. Indeed, had there been more resources directed towards developing broadly-protective COVID-19 vaccines during the early years of the pandemic, and more urgency to develop them in parallel to the first generation of Spike-based vaccines that were deployed, the many lives lost across the world as vaccine efficacy waned in the face of the viral mutations constituting the Omicron variant might have been saved.
With precisely that lesson in mind, I founded PopVax about four years ago to develop broadly-protective vaccines intended to remain effective even as pathogens like SARS-CoV-2 mutate rapidly, protecting not only against the threats of today but also the multitude of menacing variants that may emerge tomorrow. Our north star is our Million Lives Mission – to develop novel vaccines and therapeutics over the next decade that eventually save 1 million lives each year.
At the time I was contemplating starting the company in mid-2021, Indian vaccine behemoths were touting their gigantic production capabilities, but had essentially no plans to design and develop the next generation of COVID-19 vaccines. Indeed, they didn’t even update their existing vaccines to combat new COVID-19 variants like Omicron and XBB – while Moderna and the BioNTech-Pfizer collaboration in the West used RNA technology, a form of programmable medicine that makes it very easy to update the immunogen in a vaccine and use the same production process to roll it out rapidly to an entire population, Indian vaccine companies were using older technologies that were far more difficult to update.
Meanwhile, the AlphaFold2 paper had just been published in Nature, and it became clear that the same Transformer architecture that was in the process of revolutionizing artificial intelligence via the birth of the Large Language Model could also be used to design biomolecules with a combination of precision and speed that was previously impossible.
I became convinced that using a combination of programmable medicine platforms like RNA and generative AI, it was for the first time possible to develop broadly-protective vaccines – a new class of vaccine that is robust to mutation, that can anticipate and preemptively block off the insidious evolutionary pathways of viruses and bacteria, and perhaps even protect against a whole genus or family of related pathogens at once.
With all my meagre savings and $100,000 of fortuitous initial funding from the Gates Foundation, we were able to set up a small R&D operation in Hyderabad in a shared lab at the Centre for Cellular and Molecular Biology (CCMB) and demonstrate our ability to synthesize and test a simple mRNA-LNP COVID-19 vaccine and inject it into animals, successfully eliciting an antibody response. Building on a fascinating insight from MIT’s Sergey Ovchinnikov and colleagues, we were able to invert AlphaFold2 to hallucinate new protein immunogen designs intended to elicit a rare class of ‘variant-proof’ broadly-neutralizing antibodies, rapidly bringing us to the technological frontier of vaccine development.
Buoyed by this early success, we began working in earnest on a broadly-protective COVID-19 vaccine. Our work caught the eye of Ethereum co-founder Vitalik Buterin, whose Balvi fund made a deal with us – millions of dollars in contract research agreements to fund the development of our vaccine platform and production capabilities, eventually totalling $15 million+, in exchange for one critical commitment:
We’ve agreed to make our COVID-19 vaccine open-source. Once we conclude a Phase I clinical trial for this vaccine, we will release design and manufacturing information that allows others to produce and build on our vaccine, and we will not enforce any of our intellectual property rights on those who choose to do so for vaccines against any betacoronavirus, the viral genus in which SARS-CoV-2 sits. We do this because we believe that SARS-CoV-2 and its close cousins continue to pose a substantial pandemic risk – they’re already responsible for one major epidemic (SARS-CoV) and one close call (MERS-CoV) in just the past few decades aside from the big pandemic – and we’d like humanity to be prepared with better vaccine technology for next time, just as the SARS-CoV vaccine designs accelerated the development of the first generation of COVID-19 vaccines.
As Vitalik has argued in his d/acc manifesto, we believe that the capability to design, develop, and manufacture the next generation of vaccines against old threats and new must be globally distributed – that the rising world in Asia, Africa, and South America needs its own native capacity to do cutting-edge research, not just manufacture medicines invented elsewhere. During the COVID-19 pandemic, the best and most recently-updated RNA vaccines were unavailable to poorer countries – the rich world grabbed all the supply. That won’t happen again on our watch.
On our journey to developing our broadly-protective COVID-19 vaccine over the past four years, we’ve shown that this isn’t just rhetoric – we’ve demonstrated how to put it into practice. We’ve built an end-to-end platform to rapidly advance programmable medicines from concept to clinic, including:
A machine learning feedback loop to generatively design protein immunogens and therapeutics with precise functional properties
A novel RNA architecture for immunogen display able to substantially increase and broaden the antibody response elicited
Our own library of hundreds of novel ionizable lipids and thousands of formulations for LNP delivery, including our flagship 4°C-stable vaccine formulation
Our RNA Foundry in Hyderabad for in-house fast-turnaround production of RNA vaccines and therapeutics in accordance with global Good Manufacturing Practices (GMP) for biologic pharmaceuticals, meeting both US & Indian standards
Perhaps most importantly, an exceptional team that now numbers nearly 100 people from some of the top universities, research institutes, and biotechnology companies in India and across the world, with deep expertise spanning machine learning, structural biochemistry, molecular and cell biology, immunology, biologic delivery, pharmaceutical manufacturing & analytics, quality, and regulatory affairs
Our mRNA-LNP COVID-19 vaccine, PVX-001, is competitive in preclinical studies with second-generation COVID-19 vaccines from Moderna and BioNTech-Pfizer, and our next step is to take it into the clinic. We’ve received approval from the Indian national regulator, CDSCO, and the local regulator in the state of Telangana, the DCA, to manufacture phase-appropriate GMP batches of this investigational vaccine in the form of formulated bulk substance, including RNA synthesis and LNP encapsulation, with only final filling into individual dose vials to be performed elsewhere.
Clinical trial doses are now being filled by Advanced BioScience Laboratories and tested for sterility by Eurofins in the United States of America as part of our partnership with the US National Institutes of Health (NIH) – more on that later.
It’s been an eventful four years, with triumphs and tragedies, but I’m happy to report that we have test vials of our vaccine in hand, with the clinical doses soon to follow, and we intend to file for and begin our Phase I clinical trial in the first half of this year.
Taking this open-source COVID-19 vaccine into a Phase I clinical trial, and demonstrating that it elicits a potent, functional immune response against SARS-CoV-2 while remaining extremely safe, are critical steps on the long and doubtless winding road to fulfilling our Million Lives Mission, but we’re on the verge at last – for the first time, there are no technical or infrastructural obstacles in our path.
Yet as we built our platform and our COVID-19 vaccine atom-by-atom over the past 4 years, a storm gathered above us...
The perfect storm upending vaccine development
Across the world, vaccine development is slowing down. In the glorious 1960s-1980s heyday of 20th-century vaccinology, a team led by Merck’s legendary Maurice Hilleman developed and brought to licensure some of the first vaccines against measles, mumps, rubella, hepatitis A, hepatitis B, chickenpox, meningitis, Streptococcus pneumoniae, and Haemophilus influenzae, versions of which are still in use across the world today. Hilleman famously used a sample of mumps from his own daughter, Jeryl Lynn, to bring to market a vaccine against that pathogen in just four years – that was the fastest timeline from initial development to approval of a vaccine ever in the US until the Moderna and BioNTech-Pfizer COVID-19 vaccines. In comparison, the first malaria vaccine took 30+ years from conception to achieve licensure in 2015, and researchers have worked for decades on diseases like HIV and HCV without a single vaccine being approved. There has long been a desire to develop pan-influenza vaccines to resolve the effectiveness issues of existing seasonal influenza vaccines due to strain misprediction and rapid mutation, as well as provide robust protection against key potential pandemic lineages, but progress towards the clinic has been minimal over the past few decades.
Why this deceleration? The low-hanging fruit have already been plucked. Problems that can be solved with live attenuated or inactivated vaccines, simple recombinant protein presentation of an immunodominant antigen, or glycoconjugation for bacterial vaccines, have largely already been solved. What remain are pathogens such as Hepatitis C Virus, Tuberculosis, and Strep A, which collectively kill ~2 million people each year across the world, as well as tricky latent pathogens such as the ubiquitous herpesviruses EBV, HSV, and CMV, which infect a majority of the population and lie dormant for decades before reactivating to trigger serious diseases, including cancer and neurodegeneration, in millions of people globally. Existing vaccine design approaches have been unsuccessful against all of these pathogens, as well as other fast-mutating viruses like HIV, whose public health burden has been significantly alleviated by effective antivirals but now seems poised for a resurgence due to a precipitous decline in foreign aid funding, and influenza, for which the existing approved vaccines are only 30-50% effective in a given season, sometimes even as low as 6% effective (as in the 2014-15 flu season in the US).
Compounding the problem is the fact that, as with all new therapeutics, the cost of developing a new vaccine is exploding. It takes $500+ million at a minimum, and multiple billions of dollars in the median case, to get a completely new vaccine to market globally. Even though the top 3 vaccines by revenue make 5+ billion dollars/year, they’re not all that successful, and many never get approved at all – a vaccine that looks promising in the early stages can easily reveal fundamental problems in later-stage clinical trials. Sanofi, one of the world’s largest pharmaceutical companies and the market leader in influenza vaccines, recently blew almost $1 billion developing a dengue vaccine that can, in the end, only be taken safely by people who have already had dengue.
Vaccines are only dosed ~1-3 times in a lifetime, and cost much less than most other biologic medicines – they are an incredibly cost-efficient mechanism of disease reduction. Despite their relatively low cost, vaccines are often difficult to manufacture, with bespoke production processes that require labour-intensive and lengthy optimization to make them repeatable, scalable, and efficient – Merck’s Human Papillomavirus (HPV) vaccine Gardasil-9 and Pfizer’s pneumococcal vaccine Prevnar-20 are prime examples, with a single dose of either taking as much as 2+ years to manufacture and test for quality.
As such, as the cost of development rises, much of big pharma and many VCs consider vaccines insufficiently profitable to invest in, and have substantially reduced their spending on the development of new vaccines in favour of the real money-makers – cancer therapeutics (Merck’s Keytruda: $30+ billion in 2025 sales) and weight-loss drugs (Eli Lilly’s Mounjaro/Zepbound: $35+ billion in 2025 sales).
This shift away from vaccine investment has been hastened by the growing movement towards vaccine skepticism and hesitancy in rich countries like the United States, which has begun to decrease the potential market for new vaccines in high-income countries and, over the last two years, precipitously reduced the flow of public funding towards novel vaccine R&D – NIH and BARDA have cancelled over $1 billion of previously-allocated funding for vaccine R&D over just the past year. Ironically, while the uptake of vaccines in countries like the US and Canada is decreasing, those in the rising world, here in India, in Southeast Asia, and Africa, clamour for greater supply of life-saving vaccines like Gardasil, but the prices low-income countries are able to pay ($1-5/dose) are not high enough to justify current development costs. A healthy vaccine market in rich countries that are able to bear $100+/dose is the wellspring of new vaccine development, and there are signs that it has begun to dry up in a way that may be difficult to reverse in the short term.
The final ingredient to this gathering perfect storm that might uproot the enterprise of vaccine development altogether is regulatory uncertainty. Whatever one thinks of the scientific merits of what the new administration at the US Department of Health and Human Services (HHS) is doing, they are certainly sincerely committed to the cause of closely scrutinizing vaccine safety. They have begun to remove vaccines from the recommended childhood vaccination schedule, have announced that they will be tightening standards for vaccine approval, and are in the process of conducting an investigation into alleged links between vaccines and autism. The US Centers for Disease Control (CDC) has replaced almost the entire membership of the Advisory Committee on Immunization Practices (ACIP), the key external vaccine advisory body, and the new ACIP chair, Kirk Milhoan, recently told Politico that
“Americans should view the panel ‘more as a safety committee. Efficacy will be secondary.’”
Meanwhile, the US Food & Drug Administration’s Center for Biologics Evaluation and Research (FDA CBER) recently refused to even review Moderna’s application for approval of its novel mRNA influenza vaccine, mRNA-1010, which showed better efficacy compared with an approved influenza vaccine in a 40,000+ person Phase III trial, the design of which the company says was previously reviewed and found to be adequate by the FDA. Though they seem to have partially relented, the confidence of pharmaceutical developers and their investors in the predictability of the regulatory pathway has already been durably shaken.
This regulatory uncertainty creates further risks and further reduces the potential rewards of developing new vaccines, increasing the possibility of an outcome where a vaccine passes existing safety and efficacy standards, but is prevented from being approved or recommended by rapidly-changing government positions and policies that are not yet well understood or, indeed, have not yet been established at all.
Ironically, many vaccine skeptics have accused big pharma of forcing unnecessary vaccines on an unsuspecting populace to juice their profits – if more profit is the goal, these companies should probably abandon vaccines entirely, which is what they’re doing now. Financial incentives are not aligned with cheaply preventing disease, but rather with selling a steady drip-drip of doses for lifelong chronic conditions, or charging hundreds of thousands of dollars for life-extending therapies to desperate patients with no other options.
This means that the investment that new vaccine development gets today, the 2020-2022 COVID-19 sugar rush aside, is far less than it deserves given the huge public health and pandemic prevention impact that money would have – Moderna CEO Stephen Bancel, for example, has said that he is not inclined to invest in Phase III clinical trials of new infectious disease vaccines moving forward, instead doubling down on their cancer programs. This is almost the exact inverse of the funding situation for rare diseases, where straightforward approval processes and multimillion-dollar cell & gene therapy prices have seen massive investment for therapeutics intended for total populations of just a few tens of thousands of people. Meanwhile, new vaccine programs that hundreds of millions of people would benefit from are starved of funding.
If we had vaccines against all the pathogens and diseases that matter, perhaps this slowdown in vaccine investment and development would not be so concerning, but we very much do not. Consider TB (in adults), Strep A, HCV, HIV, and influenza – there are no approved vaccines for the former four pathogens, and the current generation of influenza vaccines are, as previously discussed, inconsistently and incompletely efficacious.
HIV/AIDS is a little different from the others – it is a uniquely difficult disease to develop a vaccine for, as we discuss briefly later in this piece, and newly developed long-acting prophylactic antiviral injections such as Gilead’s Yeztugo/lenacapavir are as much as 99% effective at preventing infection over 6 months, with the durability of efficacy increasing as this class of drugs is developed further, making them almost vaccine-like. The broad availability of pre-exposure prophylaxis (PrEP) has precipitously reduced both new infections (40% reduction over the past 15 years) and deaths, with the downward trend continuing apace – this isn’t true for the other pathogens in the table.
But as one pathogen is conquered, others gain strength and multiply. The combined 3 million lives lost to TB, Strep A, flu, HCV, and HIV each year now pale in comparison with the ESKAPE+E set of bacterial pathogens, 7 bacteria that are known to rapidly mutate to resist antibiotics. These antimicrobial resistant (AMR) bacteria are collectively responsible for a whopping 4+ million deaths each year, mostly in developing countries like India (700,000+ deaths/year), but increasingly in rich countries as well, including ~30,000 deaths in the US each year, largely dominated by Staphylococcus aureus and Enterococcus faecium – that is more than the 18,500 deaths/year from HIV/AIDS in the US.
With global antibiotic use having nearly doubled since 2000, the level of AMR in infections in dominant strains of these pathogens across the world is rising relentlessly, accounting for half or more of all new infections in some countries, and likely the majority of deaths. In many lower-income countries, even existing second- and third-line antibiotic treatments are rapidly losing efficacy – these bacteria are only a few lucky mutations away from rendering our most advanced antibiotics entirely useless. Meanwhile, the rate of discovery of new antibiotics is far too slow to keep up due to both difficulty and underinvestment – antibiotic development is one of the few pharmaceutical fields with even worse economics than vaccines! With apologies to William Gibson, our post-antibiotic future is already here, just not evenly distributed – we must accelerate the development and translation of vaccines, which represent an orthogonal mechanism to stop the bacteria, while our current generation of antimicrobial defenses still holds, lest we be plunged into a new dark age where a small open wound could lead to a gruesome death. Bacterial pandemics, in particular, are being almost ignored as a potential risk factor, even though the plague has previously killed 50+ million people within a single decade, and even cholera claimed 1 million+ lives over a similar span of time.
And these are just the pathogens that we already understand the consequences of! The scientific and medical communities are increasingly beginning to understand the substantial health burden associated with ubiquitous pathogens that cause chronic infections in most of the human population, often lying latent for decades without substantial symptoms. These may seem innocuous – if everyone has them, how much harm could they possibly do? – but the discovery that HPV causes almost all cervical cancer, which kills hundreds of thousands of women each year across the world, and the emerging consensus that multiple sclerosis (MS) is caused in part by the Epstein-Barr Virus (EBV), which latently infects 90%+ of adults across the world and is better known for causing mononucleosis in teenagers, have demonstrated that the long-term consequences of infection by these pathogens can be dire. There are no approved vaccines for most of these pathogens, including Cytomegalovirus (CMV – 80%+ of all adults infected), which is associated with many cancers, and the Herpes Simplex Viruses (HSV 1 & 2 – 70%+ of all adults infected), which are beginning to be implicated in Alzheimer’s disease and other neurodegenerative conditions. Many of them have no curative treatment available either – once you’re infected, you’re infected for life.
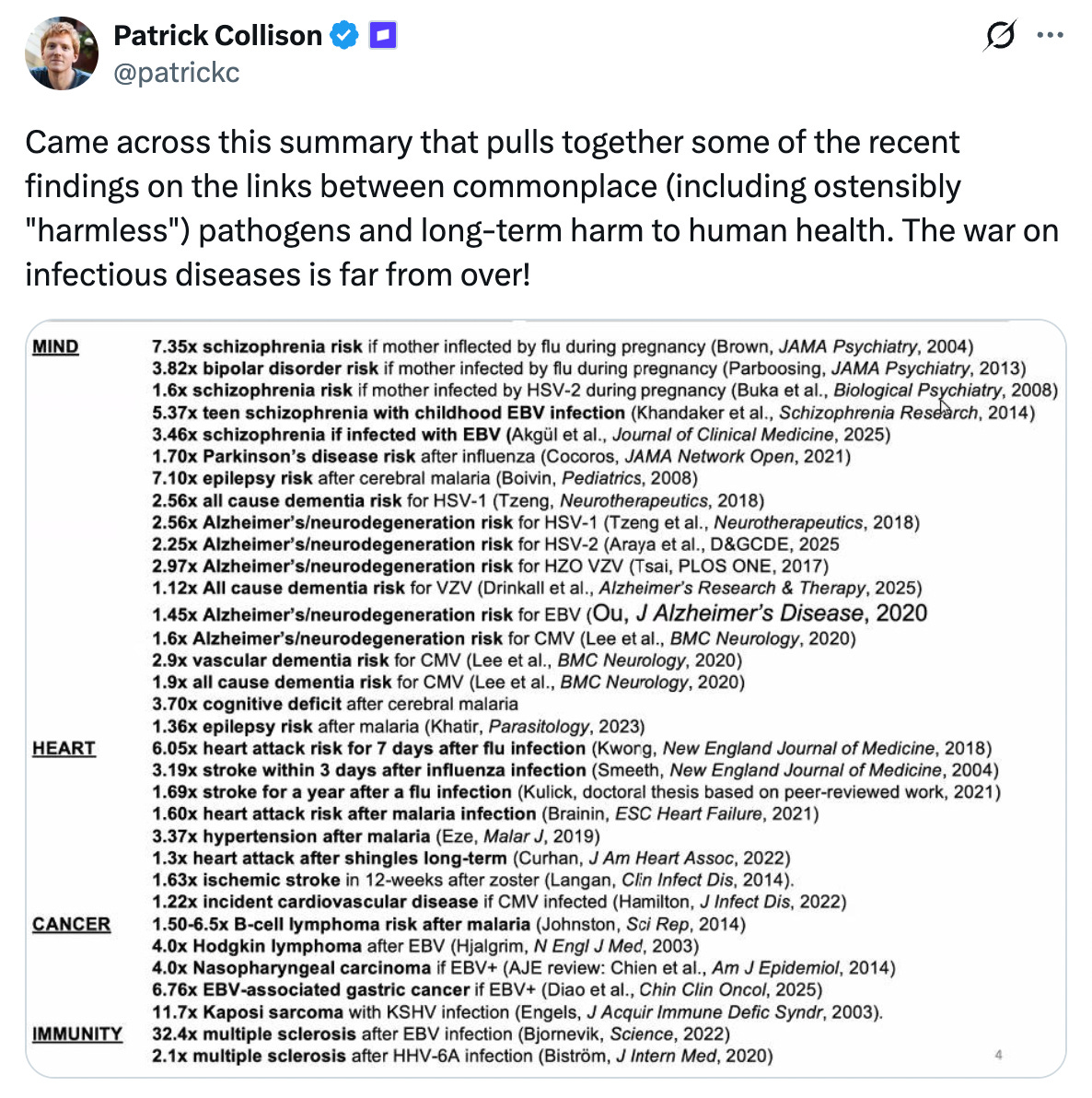
We at PopVax believe that yet more serious long-term sequelae of these infections will emerge, including cancers, organ damage, and neurological diseases, and that a greater fraction of life-threatening degenerative conditions than most people expect will be found to be downstream of latent infection. In the near future, vaccines against these pathogens will be recognized as potent longevity medicines to extend healthy lifespan, and anyone serious about their long-term health will be taking them long before symptoms appear.
Powerful proof of this hypothesis is already emerging. A recently-published natural experiment of administration of the shingles vaccine Zostavax – which prevents the reactivation of the Varicella Zoster virus, the herpesvirus best known for causing chickenpox – showed a 20% reduction in dementia risk among the vaccinated compared to the unvaccinated over 7 years of follow-up, with a similar study of the newer vaccine Shingrix demonstrating greater risk reduction relative to Zostavax. This suggests that vaccines can fundamentally shift the trajectory of neurodegeneration, and that targeting latent pathogens may be the key to preventing the chronic diseases we once thought were inevitable.
Investment in vaccine R&D has declined to the point that no major pharmaceutical company is currently prioritizing the development of novel vaccines against most of the pathogens we’ve discussed so far, with the arguable exceptions of influenza, Strep A, and EBV. This is a far cry from a decade ago, or even 5 years ago, when many of the largest pharma companies had ongoing early and mid-stage programs for many of these pathogens simultaneously. Some of these programs failed outright, because the old-school tools brought to bear turned out to be inadequate for the task, and some were rooted out over the past few years by the aforementioned perfect storm of rising development costs, a shrinking market, and sudden regulatory uncertainty.
Doubtless the winds in Washington will someday change, and the headwinds that face those brave enough to attempt takeoff in the difficult quest to develop these vaccines will give way to tailwinds that carry them to victory. In the interim, every year we let this situation continue, we condemn to death millions of people who otherwise could have been saved. If we could only find a way to accelerate, to develop these vaccines a few years, a few months, a few weeks, even one day sooner – many, many innocent lives hang in the balance.
v/acc: The era of vaccine acceleration
PopVax was founded on the belief that the combination of generative AI and new biotechnologies could open a path to saving the millions of lives still lost each year to infectious diseases by enabling the development of novel vaccines that were simply not possible with prior methods. We’re determined not just to continue on our path despite the dark clouds on the horizon, but to accelerate towards fulfilling our Million Lives Mission of developing and bringing to licensure vaccines and therapeutics that can cumulatively save 1 million lives each year.
In particular, we aim to develop highly effective vaccines to prevent TB in adults, Strep A, malaria, and hepatitis C.
Vaccinology can be a bit of a stodgy field – the fact that you’re injecting healthy people means you can’t really tolerate any safety issues, very much unlike its sister field of cancer immunotherapy. This encourages a conservative impulse in design and development, a trait that the recent regulatory shakeups in Washington will only reinforce, even though that approach certainly can’t ensure safety – indeed, in the case of dengue, conservative vaccine designs actually caused enhancement of the disease in those who were vaccinated.
At PopVax, we have the exact opposite impulse – we believe that we need step changes in vaccine technology, not merely incremental advances, to prevent the diseases we’re targeting in particular, and to confront the increasingly difficult landscape of pathogens we face in general. We’ve had substantial success leveraging emerging vaccine technologies, generating exceptional first-in-the-world preclinical data demonstrating the elicitation of deep and broad immune responses against fiendishly tricky pathogens like HCV and the fast-mutating COVID-19, but we have only begun to scratch the surface of their vast potential.
Now that we’ve exhausted the capabilities of time-tested approaches, this is the exact moment to double down on new technologies that can buttress the bridge we must build from the era of Hilleman, over the troubled waters of our stormy present, and into our glorious future in which no man, woman, or child need fear infectious diseases.
Here’s our technical roadmap for how we get there:
RNA reloaded: the nucleic acids return
Given the saturation coverage they received at its pandemic-induced peak, RNA vaccines may already seem old hat, even passé in these fast times, but it’s worth diving deeper into why the platform has the potential to revolutionize vaccine development and manufacturing.
Consider the variants. For most of the COVID-19 pandemic, India was relying entirely on two locally-manufactured vaccines for our immunization capacity – the Oxford-AstraZeneca viral vector vaccine, made and marketed by the Pune-based Serum Institute of India as Covishield, and Covaxin, an inactivated virus vaccine developed and manufactured by the Hyderabad-based Bharat Biotech with the support of the Indian Council of Medical Research (ICMR).
In September 2022, Moderna and BioNTech-Pfizer both released bivalent boosters containing mRNA encoding the Spike antigen from both the wild-type version of SARS-CoV-2 (as was in the original vaccines) and the Omicron BA.5 subvariant, and deployed them across the US and Europe. Meanwhile, here in India, as COVID-19 cases began to rise again at a worrying rate in lockstep with the rise of immunity-escaping mutations, there was no serious attempt to release new variant-adapted versions of the Oxford-AstraZeneca/Covishield vaccine or the Bharat Biotech Covaxin vaccine.
So why were Moderna and BioNTech-Pfizer able to both scale their manufacturing processes more quickly from 2020-2021 and then (in 2022) very rapidly modify their processes for variant-adapted boosters, while Bharat Biotech and the Serum Institute of India weren’t?
This comes down to the fundamental nature of the RNA platform. All viral vaccines show the body key viral proteins that elicit protective immune responses – these immunogens train the body to fight the target virus with antibodies and T cells if and when it ever makes an appearance. Most viral vaccines accomplish this by either culturing some version of the virus itself at a large scale to directly inject (Bharat Biotech’s Covaxin falls in this category), or by producing only the relevant key viral proteins in cell culture, in both cases requiring growing cells at the scale of tens of thousands of litres in giant bioreactors. These production processes are held hostage by biology – cell culture of that magnitude requires massive, purpose-built facilities and is inherently finicky in ways that defy stabilization, resulting in a long and slow scale-up and optimization process to coax the cells to reliably produce the virus or proteins. The pharmaceutical researcher and blogging eminence Derek Lowe said it best in this excellent Chemistry World piece on these difficulties – “Human cell culture is voodoo. Cells do what they want.” Each batch starts from a small-scale culture and goes through phases of scale-up in volume over the course of 30-40 days until it is ready for harvesting, and you might get wildly different results from batch to batch.
RNA vaccines sidestep this bottleneck. Messenger RNA (mRNA) is the molecular code that instructs ribosomes in human cells to synthesize specific proteins – we commandeer this mechanism to instruct the body to produce the key viral proteins that we want to train the immune system to recognize and fight.
RNA is produced via in vitro transcription, or IVT, which is performed using enzymes in largely cell-free fashion from a tiny amount of template DNA produced in bacterial cells – that is, the RNA is not grown in cells but rather synthesised in a simpler, free-floating biochemical reaction. At PopVax, we perform IVT in an integrated, ‘one-pot’ reaction, and it is accomplished over the course of a single day, unlike the 30-40 day cell-culture process, massively speeding up process optimization. This is far less finicky than multi-phase cell culture, since no living organisms are involved and there are far fewer independent steps. It is also far more space efficient – the number of doses worth of RNA we can produce in a 250-litre vat would require many thousands of litres of cell culture in giant floor-to-ceiling tanks, all of which massively simplifies scaling in terms of time and resources.

On the customizability side, this means it is far simpler and faster to switch out the coding sequence of the RNA produced by just changing the corresponding sequence in the DNA template used in the reaction – any issues can be worked out over a few batches over just a few days, rather than over many months.
RNA’s speed of process development and execution is what enabled Moderna and BioNTech-Pfizer to get their COVID-19 vaccines into clinical trials only ~2 months after starting to design them, and get emergency approvals for wide distribution in just ~11 months total, shattering the long-held development-to-approval timeline record of Maurice Hilleman’s Jeryl Lynn strain mumps vaccine, which took 4 years end-to-end. They were able to get subsequent variant-updated boosters produced and approved on even shorter timelines.
AstraZeneca’s COVID-19 vaccine actually uses the same mRNA mechanism to instruct cells to produce the viral proteins, but it uses a chimpanzee virus, thought to be safe in humans, to produce and deliver DNA into the cell nucleus that is then transcribed into RNA by the cell. This vaccine platform is thus also subject to the vagaries of cell culture to produce this virus at a large scale, while also eliciting immune responses to the chimpanzee virus carrier that may compromise the effectiveness of re-dosing and boosting – in many ways, the worst of all possible worlds. As such, changes to the COVID-19 sequences being cultured substantially disrupted the stability of both the Covaxin and Covishield manufacturing processes, requiring many months of painful re-optimization that rendered rapidly updated boosters chronologically and economically infeasible.
With RNA, on the other hand, it is even possible to go from a newly designed vaccine sequence to full-scale GMP production in as fast as a week if there is a pre-existing process in place, though of course quality control testing of the final product would typically take longer than that. If accompanied by a fast generative AI-assisted design process, one could rapidly churn out dozens of novel vaccine candidates a year from a single facility to fight the world’s most destructive diseases, not to mention new pathogens that may soon emerge from the underground labs of AI-assisted bioterrorists at lightning speed.
For the same reasons, it is much, much faster and easier to manufacture a highly-multivalent vaccine using this platform featuring multiple different RNAs, as compared with current examples such as Gardasil-9 and Prevnar-20 with their aforementioned multi-year manufacturing cycles. Centivax, a San Francisco-based vaccine startup, leveraged this multivalency advantage to develop a potentially universal flu vaccine candidate encoding Hemagglutinin (HA) antigens from 22 different influenza strains to solve the efficacy challenges that we face with current vaccines – their candidate, which entered clinical trials just last month in Australia (February 2026) would likely have been impractically complex to manufacture using the egg- and cell culture-based production platforms of conventional flu vaccines.
In essence, RNA opens the door to testing a far more ambitious range of vaccine candidates across a far broader range of diseases than is financially or logistically viable with other platforms. The platform itself – the expression, delivery specificity, stability, and safety profile – can keep improving in parallel, and each new candidate gets the benefit of the latest set of improvements without needing to reinvent the wheel each time. This is the future of vaccines – an ever-improving platform, fast candidate iteration, and compact production, all resulting in a near future hopefully far less troubled by pathogenic mortality than we are today.
All of this may seem like conventional wisdom for those of you who watched the news in 2021, and you might reasonably wonder if this is all being taken care of already by big pharma – Pfizer, Moderna, BioNTech and their ilk, the players that dominated conversations about mRNA vaccines at the pandemic’s peak, but as these large companies pull back from building infectious disease vaccines on their RNA platforms for the reasons we previously discussed, and US government funding for RNA evaporates, the future of RNA vaccine R&D, which is essential to defeating our pathogenic enemies, may lie in unexpected places rather than the conventional corridors of legacy biotech power.
Sitting here in India, at the heart of the rising world, we stand ready to make a long-term bet on what RNA vaccines can accomplish. To have any chance of winning this bet, we must and will improve the technology to mitigate and eventually eliminate the low-frequency but well known cardiac safety issues seen in a tiny fraction of the billions of people who have already taken them, and we must do this as transparently as possible to earn and keep the confidence of our fellow people of earth. We believe that this is possible over the next 5 years, and that solving these problems will make RNA the default choice for many, many types of vaccines in the long run.
Unorthodox programmable immunogen presentation methods
A lot of the new vaccines introduced over the past two decades, including the HPV and malaria vaccines, are based on virus-like particle (VLP) display of immunogens to substantially boost immunogenicity. This involves injecting an assembly of multiple copies of an immunogen that form a ball-like structure, similar to how the virus would look in the body in its native form, that enables repeated display of key epitopes, the minimal immunogenic motifs that elicit antibodies and T cell responses, including epitopes formed at the interface of multiple copies of the immunogen.
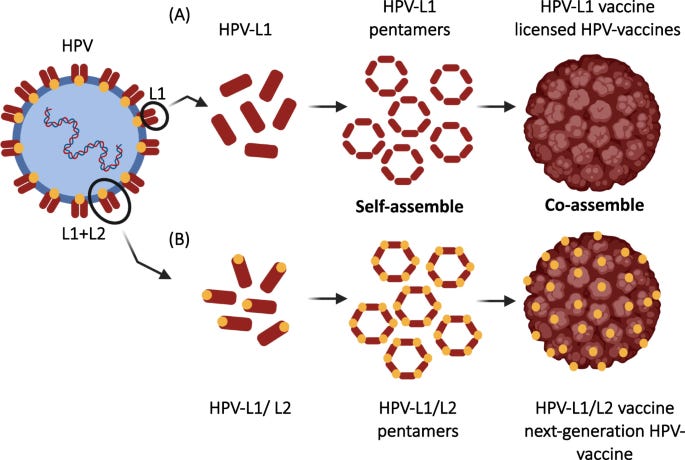
In the case of the HPV vaccine, above, this is a natural property of the HPV L1 protein, so it is directly used as the immunogen. In the case of the malaria vaccines, the Plasmodium falciparum Circumsporozoite Protein (CSP) immunogen used does not have this property, so they fuse it to the Hepatitis B surface antigen (HBsAg), which does, enabling the fusion protein to self-assemble into a VLP displaying part of the CSP.
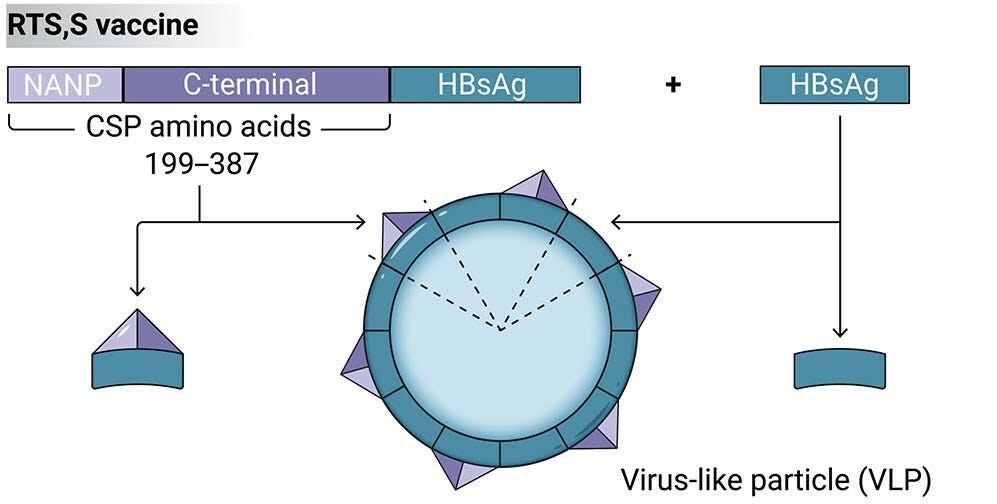
The problem is that VLP vaccines are complex to develop and manufacture, especially when there are multiple pathogen genotypes to be covered, as in the case of HPV – though L1 self-assembles in the body during the actual viral replication process, externally assembling VLPs of each of the 9 different genotypes in current HPV vaccines required lengthy and subtle process optimization, even after which each dose likely takes 2+ years to manufacture and quality control end-to-end, as previously mentioned. This complexity is why VLP vaccines for many pathogens, where the technology may help boost efficacy or reduce required doses, haven’t yet been advanced far down the clinical pipeline. This is particularly true for pathogens like COVID-19 and influenza, where it may not be possible to update these manufacturing processes quickly enough to accommodate annual strain changes.
PopVax has pioneered an RNA-encoded VLP platform to get the best of both worlds – the efficient and fast manufacturability of mRNA-LNP vaccines, including the ability to rapidly update immunogens, alongside the efficacy boosts that VLPs offer. Our approach is to inject an RNA-LNP that translates and self-assembles in the body into a VLP displaying the immunogen.
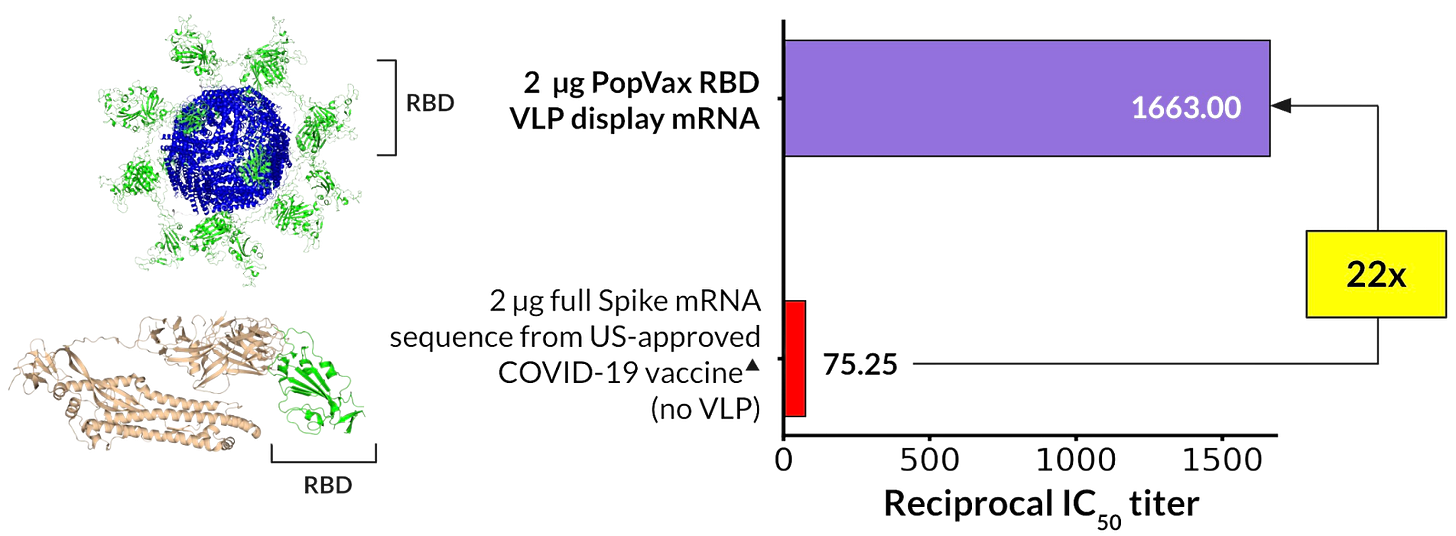
When applied to the design of a wild-type COVID-19 vaccine, our RNA-encoded VLP approach results in a >50x increase in the elicited neutralizing antibody (nAb) titer in mice, as well as a >20x increase in nAb titer compared to the same dose of the mRNA sequence from a US FDA-approved first-generation wild-type COVID-19 vaccine. Our wild-type SARS-CoV-2 RBD-based VLP display construct was also able to elicit potent neutralization titer against the Gamma, Lambda, and Omicron BA.1 variants of COVID-19, while the FDA-approved mRNA-based COVID-19 vaccine comparator sequence at the same dose was not, demonstrating that the VLP immunogen display approach results in the elicitation of antibodies that neutralize a greater breadth of variants – this remarkable property is of great relevance given our desire to develop a broadly-protective COVID-19 vaccine.
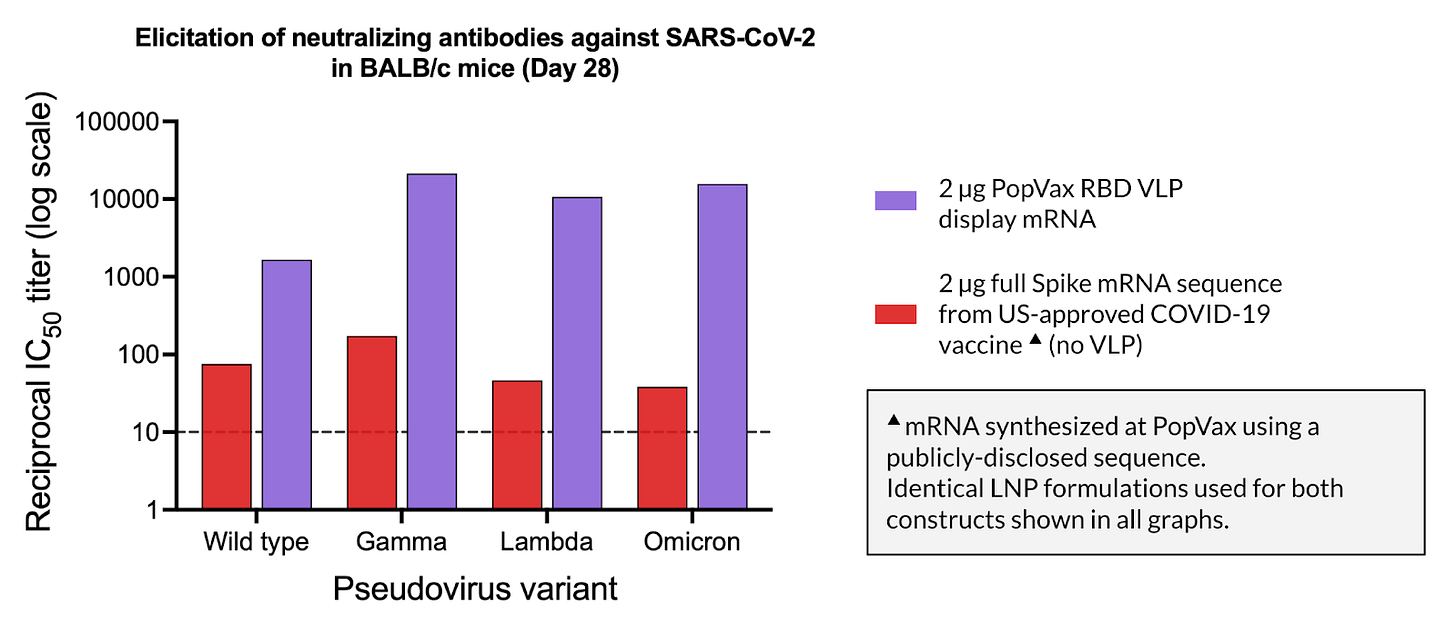
Our current COVID-19 vaccine candidate, PVX-001, which will enter clinical trials this year, uses our RNA-encoded VLP platform to display an engineered version of the JN.1 variant RBD, and is able to potently neutralize all recent and present prevalent variants, including KP.2 and LP.8.1, the latter of which was dominant for most of last year. Our preclinical immunogenicity in mice is competitive with Moderna’s much improved second-generation vaccine, mNEXSPIKE, which uses a different immunogen engineering and display approach from the original.
Beyond RNA-encoded VLPs, we are working on other novel methods of immunogen presentation, including for bacterial vaccines. Many bacterial vaccines, such as Prevnar, rely on immune responses to polysaccharide antigens, which are difficult to rapidly manufacture or modify in response to bacterial mutations, and require specialized equipment and production methods. Using generative AI and high-throughput screening methods, we can develop protein mimics deliverable via programmable RNA-LNP platforms that can be modified and manufactured on short notice in general-purpose facilities. We are also exploring T cell antigen presentation methods that are independent of Human Leukocyte Antigen (HLA) subtypes, which vary widely between individuals – this would help increase the breadth and potency of vaccines against intracellular bacteria and parasites, which have historically had mixed effectiveness.
Designing better delivery for programmable vaccines
RNA-LNP vaccines are both extremely fast to develop and update, and are also by far the most convenient to manufacture – a facility capable of producing 1 billion doses each year can have a footprint as small as 100,000 square feet, and even our RNA Foundry, with under 5,000 square feet of GMP manufacturing space, can produce up to 1 million doses each year. Lipid nanoparticle delivery, however, is far from perfect – the current generation of LNPs:
tend to cause more noticeable short-term reactions than other vaccines, including soreness at the site of injection, chills, fatigue, and even low-grade fever
are expensive to transport due to cold-chain requirements, with many requiring supercold (-20°C) storage as opposed to the more common standard 4-8°C refrigerator temperature required by most other vaccine types
are often running on empty – a lot of LNPs in a vaccine vial don’t have any RNA in them, and are thus just increasing the cost and side-effects of an RNA vaccine without adding any efficacy
Better LNPs that optimize these properties would increase the safety and reduce the cost of RNA vaccines. We have already developed a 4°C-stable LNP formulation based on our novel ionizable lipid PVXL-150 that shows high tolerability and minimal side effects in preclinical models, which we will soon be advancing into a Phase I clinical trial as part of our first vaccine program this year.
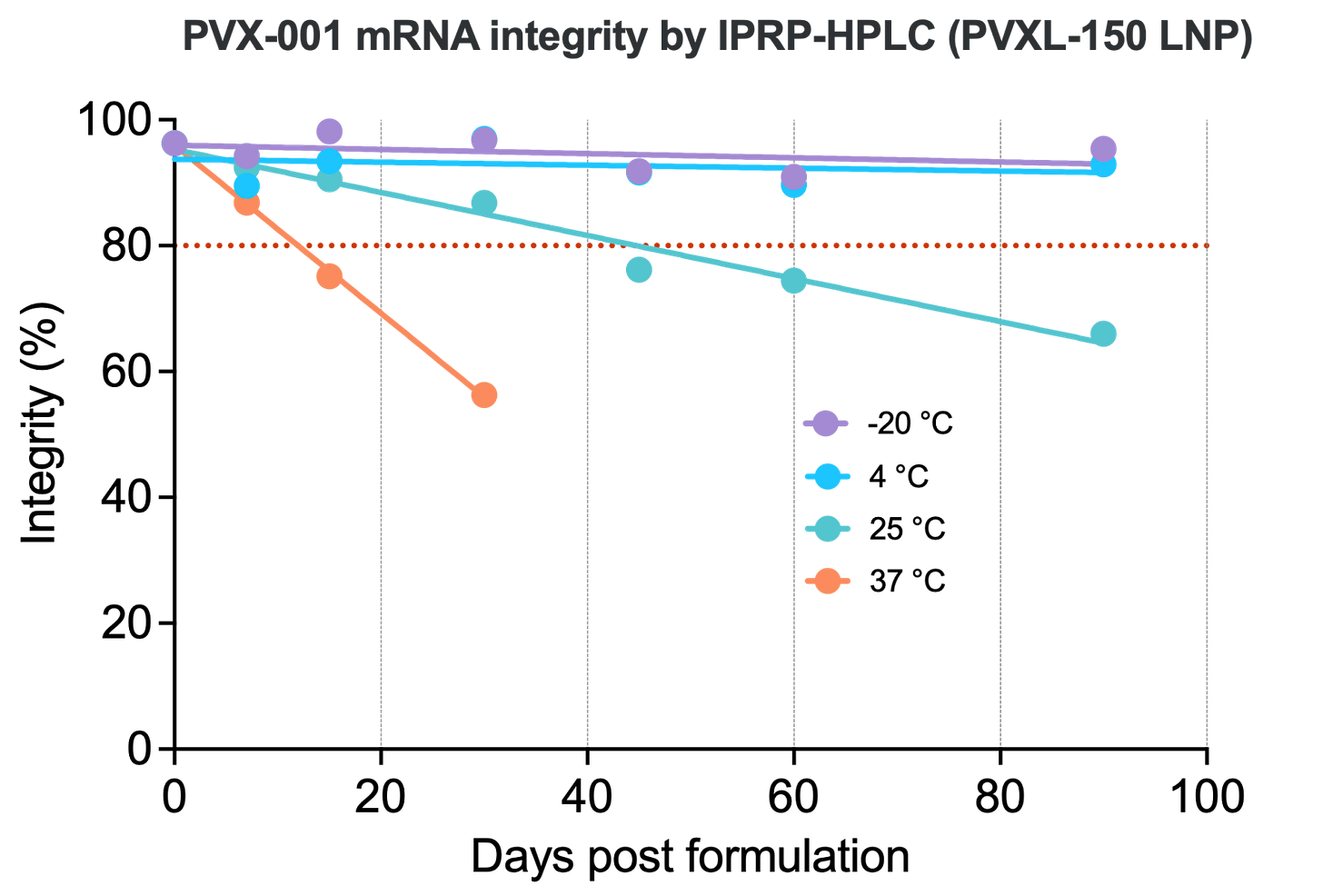
We are now producing large, high-quality datasets linking lipid and LNP design to functional properties, including immunogenicity, tolerability, and stability, which will allow us to train generative AI models to design highly-effective and safe LNPs.
Better LNP chemistry aside, we’re also working on ways to dramatically simplify vaccine deployment. To receive a vaccine today, you have to go to a clinic, wait in line, and be injected by a trained professional – a logistical bottleneck that, as we saw at the height of the COVID-19 pandemic, forced millions of people to queue up in crowded clinics to receive a vaccine against a virus that spreads in crowded rooms.
It doesn’t have to be this way. Imagine a patch, the size of a thumbnail, delivered straight to your door via UberEats, Zomato, or Doordash, that you press to your skin for fifteen minutes to administer the vaccine – no clinic, no needle, no queue! We were awarded $2 million by BARDA, the US biodefense agency, as part of their $50 million Patch Forward Prize to make this vision a reality in partnership with Germany’s LTS Lohmann, and we’re working on it as we speak. As a bonus, formulating an RNA-LNP vaccine into a microarray patch may further improve its thermostability, potentially removing the need for refrigerated storage and transportation altogether.

Immune Organoids: bringing the human immune system into the lab
Mice don’t get AIDS. They don’t get malaria, hepatitis C, or tuberculosis in any way that resembles how humans do. For decades, this inconvenient biological fact has been papered over by a combination of genetic engineering – humanised mice, transgenic mice, mice with deliberately compromised immune systems – and a collective willingness to pretend that a response in a rodent tells you something reliable about a response in a person. Sometimes it does. The COVID-19 vaccine generates a potent immune response in mice, which is why we use them for immunogenicity studies. But mice don’t meaningfully get sick from SARS-CoV-2 infection, because their ACE2 receptor differs sufficiently from the human version that the virus cannot efficiently bind and enter mouse cells, which is why infection models required a different animal. In the case of SARS-CoV-2 these were golden hamsters. That’s two different species, two different models, just to study one pathogen.
But the problem runs deeper than just pathogen susceptibility. The mouse and human immune systems diverged over 65 million years ago, and they have spent that time in different habitats, solving different problems by evolving different apparatus. As a result, humans possess a distinctly different repertoire of antibody and T cell receptor (TCR) germline sequences than mice. These sequences, created via the recombination of V(D)J gene segments in B cells (for antibodies) and T cells, are the progenitors of every antibody and TCR the immune system produces. A vaccine doesn’t bring forth new antibodies from scratch – the immune system selects, hypermutates, and expands B cells that already exist within this library in response to the immunogens in the vaccine. A vaccine can only elicit a particular antibody if the right precursor clone exists in the repertoire of the person receiving it – a rare broadly-neutralizing HCV antibody found in humans who naturally clear the infection, for example, may simply have no equivalent in any naturally occurring mouse species. Mouse germinal centres are also smaller and more transient than human ones, giving B cells fewer cycles of mutation and selection to converge on high-affinity clones even when the right precursors are in place.
Layering onto this complexity is the fact that every human immune system is tuned differently. T cells, for example, do not work on the level of whole immunogens – instead, they recognize short peptide fragments of them. The molecular machinery that decides which peptide fragments of the immunogen get flagged for attention, the Major Histocompatibility Complex (MHC), varies enormously across people, but remains nearly invariant across inbred laboratory mice. B cells present these peptides, mounted on MHC molecules (known as HLAs in humans), to T cells, and these B cells receive the signal to expand and eventually produce a large amount of antibodies only if T cells bind to the specific MHC-bound peptides it is displaying. Put the same immunogen into a mouse and a human, and even if the right B cell precursor to elicit a potent neutralizing antibody exists in both species, the actual antibody may only be elicited in one, the other, or neither, based on whether or not each organism has the right MHC subtypes to display peptides from the immunogen that activate CD4+ T cells – you would also see this variation from one human to the next.
The consequence of all of this compounding complexity is straightforward: an immunogen that elicits a massive functional immune response in a BALB/c mouse may barely evoke a whisper of a response from a human immune system, and vice versa. Conventional vaccines using large immunodominant immunogens that include many potent overlapping epitopes often mask this reality, since something will likely elicit a response in any given species, but a precision vaccine using specific epitopes to elicit a specific immune response – precisely the kind of vaccines most likely to be able to make a dent in long-unsolved problems in vaccinology for pathogens like HCV – will fall right into the translational gap.
Don’t panic! The answer to one of vaccinology’s most perplexing problems may be sitting in ENT surgeons’ waste bins. Tonsils, which are routinely removed and discarded as waste, are secondary lymphoid organs – the same class of immune organs as lymph nodes and the spleen. Cells derived from these specimens can be cultured outside of the body to reconstruct a functioning human lymphoid environment – a microcosm of the human immune system – in a petri dish. Mark Davis at Stanford and Lisa Wagar at UC Irvine have demonstrated that these immune organoids capture the key features of a human adaptive immune response to a vaccine antigen, including B cell activation, maturation, and differentiation into antibody-secreting plasmablasts, in a way that no mouse or other animal models can reliably replicate. In simple terms, these miniaturized immune systems produce antibodies in response to vaccines that look like the ones you and I would produce.
PopVax is adapting these protocols to enable the rapid functional testing and iteration of novel vaccine designs. We aim to bridge the translational gap between the lab and clinical immunology without putting human lives at risk.
Predictive & generative AI models of human immune responses
Generative AI for protein design has gone from being a theoretically interesting notion in 2020 to very real and very useful in 2026 – hit rates for fully de novo designs with sophisticated pipelines now routinely exceed 20%. It is now possible to design a precision vaccine to display a specific epitope in mere hours, a task that previously might have taken the length of an entire PhD with physics-based methods. PopVax is one of the first vaccine developers to be AI-first from day one – our aim since founding has been to leverage the power of machine learning to design vaccines to elicit specific antibody and T cell responses against pathogens that prior vaccine design approaches have been unable to overcome.
What we’ve learned over 4 years of doing this is that, though generative protein design models have gotten better and better at an accelerating pace, and they now have the ability to design proteins that reliably recapitulate the desired epitope structure, as well as proteins that can bind with specific antibodies that one wants to elicit, they aren’t yet particularly good at designing effective immunogens that are actually able to elicit the desired immune response.
This is because how the immune system is going to respond to a pathogen, vaccine immunogen, or therapeutic protein is still largely a black box to us – despite substantial progress in immunology, we don’t really know what the specific antibody or T cell response is going to look like beforehand. The actual protection conferred by a vaccine is a second-order effect – you design an immunogen that doesn’t directly protect from a pathogen, but rather stimulates the immune system to elicit antibodies that are, in turn, hopefully protective. We need to test each immunogen in animals or in the immune organoid models discussed above to see if the immunogen works at all or does any better than what came before. Designing immunogens to elicit precise but potent immune responses to epitopes beyond those that are already immunodominant is just barely possible via slow, iterative validation and optimization via animal or organoid immunization, with each immunogen taking many, many cycles, with each often months long, to get right.
Armed with our immune organoid models, PopVax is developing ways to rapidly collect human-relevant immune response data for hundreds, thousands, and eventually hundreds of thousands of novel AI-designed immunogens, which we will then use to train predictive models mapping immunogen design to the elicited antibody and T cell response in humans. Once we have predictive models that work, we can turn those into generative models that will allow us to design immunogens conditioned on and intended to elicit specific antibodies or T-cell responses we want to elicit.
The compounding advantages of fast, cheap experimental data
PopVax is a glimpse at the future of vaccine development. This future will be unevenly distributed, just as the recent past has been.
Since the founding of Genentech in South San Francisco, California, almost exactly half a century ago, and the opening of Biogen’s labs in Cambridge, Massachusetts, a few years later, biotech inexorably gravitated towards these hallowed lands, a galaxy of startups and goliath pharmaceutical companies orbiting these twin blazing beacons. Genentech, Roche, Genzyme, Sanofi, Biogen, Johnson & Johnson, Vertex, Gilead, Millennium, Takeda, AbbVie, Pfizer, Alnylam, Calico, Ginkgo, Novartis, Vaxcyte, Moderna – they all lined up scientists cheek by jowl at water’s edge, convinced that the world’s most innovative new medicines could only be developed within these few square miles.
Venture capitalists backed them enthusiastically – SF and Boston combined routinely account for over 40% of all biotech VC dollars raised in a given year across the world, just a few zip codes taking on the collective R&D might of the rest of the planet and holding their own. Harvard and MIT professors often spun out startups based on little more than an interesting academic paper that were handed $25 million on day one, and for decades big pharma eagerly scooped them up for a billion or two at the first sign of positive data for their lead drug candidate.
And if this approach meant that some types of medicines, including many vaccines and nearly all antibiotics, fell by the wayside because the low prices their developing-country markets could bear couldn’t justify US development costs, well – that was just a fact of life. Sure, it might take a few billion to get a new drug approved these days, but you can’t put a price on good science, can you?
It turns out, you can, and it can be a lot cheaper! Just like their midwestern countrymen a generation before, Boston and SF never saw China coming. Sure, they all increasingly relied on Chinese contract research organizations (CROs) like Wuxi to run their actual wet lab experiments, and Chinese contract manufacturers to make their experimental medicines, but these were just unthinking service providers, right? They spun stories about how China was cheap but unsophisticated, and that US biotech would always be a shining city on a hill, far above the madding crowd.
Meanwhile, Chinese biotech companies put their heads down and built a drug discovery engine from scratch – they’ve gone from essentially 0 to a third of major global pharmaceutical licensing deals over just the last decade (per Bloomberg). Chinese companies now originate more novel molecules than all of Europe combined.
For some insight into how that happened, it’s worth reading Alex Telford’s seminal piece, Will all our drugs come from China?
“If there’s any single factor that matters the most, it’s probably that Chinese companies are just faster. Legend was founded in 2014, got into clinical trials in 2016, and did its deal with J&J in 2017. By contrast, it usually takes Western biotechs 3 to 6 years to satisfy IND requirements and get a new drug into the clinic.
Western biotechs that discover promising new targets for intractable diseases are finding that Chinese teams take their published ideas and beat them to generating clinical data.”
Informed by our cultural learnings from Chinese biotech, PopVax plays the sport of biotech R&D with an entirely different strategy than our colleagues out west. Drug discovery is inherently stochastic – no matter how much of a genius you are, and no matter how earth-shatteringly brilliant your therapeutic ideas, most of them will die an ignominious death from a thousand cuts inflicted by the fractal complexity of human biology, of which we still understand less than we don’t (recall, for example, the section on antibody elicitation above). Maximizing the odds of success is a game of quantity – we have to get as many high-quality shots on goal as fast as possible and as cheaply as possible.
That isn’t hard to do when your scientists are just as good, some fresh off the plane from this Cambridge, or the other one, and a typical experiment in our wet lab costs as little as a tenth of what it does in the US. Bespoke infrastructure can be built in months for what amounts to pocket change on the scale of biotech R&D – even building a customized GMP manufacturing facility has been at least 5x cheaper than we imagined.
We turn that cost advantage into a speed advantage – we can test 10x more hypotheses, 10x more potential medicines, in our lab per dollar of funding we receive. That parallelism, those shots on goal, mean that our time to preclinical success, how long we’re waiting before any one thing works at all, shortens dramatically – we’re rolling 10 dice a turn.
In past eras, that might not have mattered all that much, since designing a single precision vaccine candidate that isn’t just a copy of the pathogen itself took teams of people years of effort – whole PhD theses worth of long hours tweaking this amino acid and that, years of slow, iterative experiments in animals – but generative AI has now massively shrunk the time and cost of protein design, producing thousands of qualified candidate sequences in mere hours. The time and cost of testing these candidates in the wet lab – in the test tube, in mice, and in organoids – is the bottleneck stemming the flow of vaccine innovation into the clinic.
PopVax is built to open this bottleneck out into a gigantic gushing geyser of high-quality data from testing these designs in biologically-relevant models, as close to humans as we can get these things to be in a petri dish. This data is worth its weight in gold. Unlike the large language models that power ChatGPT, Claude, and Gemini, today’s generative AI models for biological design are bottlenecked by data, not compute – most of them can be trained using as little as 10 to 100 GPUs, because there are often just a few hundred thousand data points to train on in the first place.
Everything about our pipeline is optimized for speed and throughput. Not only does generative AI compress our design cycle from months to hours, but our secret sauce is that using RNA as our platform of choice means we can produce hundreds of computationally-designed vaccine immunogens simultaneously in mRNA-encoded form and test them in mice or immune organoids, generating functional immune data at scale, rather than manually designing only a few candidate immunogens and painstakingly producing and purifying each in cell culture. This buys us the luxury to treat a failed experiment as data rather than a sunk cost, allowing us to assemble the most functionally-relevant machine learning datasets to train and fine-tune our generative models faster and cheaper than anyone else, creating a feedback loop to rapidly improve and optimize our immunogens.
Our in-house GMP facility sits just through the glass from our R&D lab at the RNA Foundry, allowing us to seamlessly translate a promising candidate from lab bench to clinical-grade batch in weeks, not years, enabled by the inherent adaptability and efficiency of the RNA-LNP platform.
Speed of design. Speed of data generation. Speed of iteration. Speed of GMP translation. In essence, PopVax is a tightly-integrated machine that turns AI-designed proteins into wet lab datasets into clinical trial batches at speeds previously thought to be impossible.
These advantages compound because each round of experiments leaves us with more task-specific data that good biological models need. These models then become better at proposing credible candidates, and then these are pushed straight back into high-throughput assays. The results feed directly into the next cycle. Slowly at first, then all at once, the process stops feeling linear. The models get better, the candidates get better, and the timelines that usually stretch discovery out over years begin to compress.
Meanwhile, our cost advantage persists all the way into the clinic – a Phase I clinical trial in the US is $3-4 million, and can be as low as $100,000-300,000 here in India! This asymmetry means we can get first-in-human clinical data for 10 candidates in parallel for the cost of a single US trial – more shots on goal & higher overall odds of success, of course, but even the candidates that fail will provide excellent data to better align our models with human physiology, and produce better and better candidates over time.
Our strategy is not built on abstract admiration for lower cost – this isn’t an Infosys-style outsourcing play – but on the understanding that in a stochastic field, one wins not by pretending uncertainty can be abolished but by building systems that can absorb it. Make enough high-quality attempts, at sufficient speed, at low enough cost, and that uncertainty begins to work in your favour. In a field where most new medicines fail, the structural advantage belongs to whoever can afford to fail fastest without flinching.
We aren’t cowed by the chaos of full-stack biology. We embrace the chaos. We’re from India – we are the chaos.
PopVax started out with a handful of employees, a few good ideas, and a CEO who knew nothing about biotech. Over the last four years, spending ~$20 million in total, not a single dollar of which was VC money, we’ve designed over a million candidate proteins using frontier machine learning methods and generative AI models, screened over 4,000 unique LNP formulations with our own library of novel lipids, and injected over 1,200 unique vaccine constructs into more than 8,000 rodents, generating vast amounts of immunogenicity data. Each experiment allows us to improve our candidates – every expression test, every ELISA, every neutralization assay tightens the feedback loop and raises the ceiling of what the next round of candidates can achieve.
In just a few months, we’ll enter clinical trials with our first program, the broadly-protective COVID-19 vaccine PVX-001, and we have 8 other active vaccine and immunotherapy programs against different targets at various stages of preclinical development. In contrast, a prominent west coast vaccine startup in the US, which also established a new vaccine platform, took double the time (8 years) and over 10x more money (~$300 million) to get their first program into Phase I trials a few years ago, with only a single other disease target in their preclinical pipeline at that time. The difference in return on investment (ROI) has the potential to be absolutely staggering.
PVX-001 isn’t just a me-too vaccine. Our preclinical data demonstrating that our RNA-encoded VLP approach and computational modification of the RBD resulted in broader neutralization across current and (at the time of design) future COVID-19 variants caught the eye of the National Institute of Allergy and Infectious Diseases (NIAID), part of the US National Institutes of Health (NIH). They offered to conduct and sponsor the Phase I first-in-human trial of PVX-001 in the US as part of Project NextGen, a US government initiative to rapidly translate innovative new vaccines and therapeutics against COVID-19 and other public health threats from the lab into wide availability.
We’ve worked closely with NIH to get their advice on our planning and execution of the toxicology, biodistribution, and immunogenicity studies in mice and rats necessary to take our candidate to Phase I in the US, and we’ve also benefited greatly from their expertise in the documentation of the manufacturing and quality control processes for novel vaccines. Our approach across all areas has received positive pre-IND feedback from the FDA over the past several years, including most recently early this year. After we manufactured the RNA-LNP material for our clinical batch, NIH worked with their partner, ABL, in Rockwell, Maryland, to fill-and-finish the final clinical vials of PVX-001 for the Phase I trial. We were confident that we were collectively in a position to file an IND for NIH to begin the trial in May of this year.
Unfortunately, we were informed last month that NIH will no longer be funding and conducting the PVX-001 Phase I clinical trial due to shifting priorities in the new US government. We have nothing but the utmost respect for and gratitude towards their team, and of course they are well within their rights to make this decision, which was not made due to any safety or efficacy concerns with our candidate.
We’re resolved to take this candidate into the clinic this year, and we’re not going to let this setback deter us. We’ve already begun the process of shifting the Phase I trial over to Australia with our own funding, where we expect that we can still inject the first patient in late May or early June. Once that trial is concluded, we will release the entire open-source design, and manufacturing package for the world to build on, as we’ve agreed with Vitalik and Balvi.
PopVax was brought to life in response to the failure of existing vaccines to combat COVID-19 variants, and the failure of Indian vaccine companies to develop anything better. With our broadly-protective COVID-19 program on its way to the clinic, we’ve come to the end of the beginning. What comes next will shake the foundations of vaccine development and, if we get this right, save millions of lives each year.
As we’ve previously detailed, vaccines are one of the most efficient technologies devised for reducing human suffering: a product that typically requires only one to three doses in an entire lifetime, and repays that intervention at civilizational scale. They do not merely prevent disease – they prevent the toil of long hours in the ICU, the tears shed at the funeral, the unbearable pain of losing one’s children, and the long tail of economic damage that trails illness across families and countries. The technology is extraordinary, but the economics of developing it in the West are, increasingly, untenable.
And so the contradiction at the heart of modern biotech has hardened into something almost absurd: the interventions with the capacity to save the most lives are often the least compatible with the financial architecture used to build them. Diseases with diffuse purchasing power, complex biology, or a burden concentrated outside rich countries are not usually starved of importance; they are starved of economic legibility. They do not fail the test of need, they fail the test of investor math. And so, year after year, the same pattern repeats: the biggest burdens attract the weakest pipelines; the hardest pathogens inherit the thinnest balance sheets; the diseases that kill the most people remain, structurally, the least funded.
At PopVax, we’ve proven that we can develop and advance a leading-edge vaccine on a novel platform into Phase I trials for a fraction of what it would cost in Boston & SF. If we extrapolate forward towards approval, we may be able to radically bend the cost curve of novel vaccine development from $1 billion per program down to just a few $100 million, with that number dominated by the cost of late-stage clinical trials.
Suddenly, the math changes. A whole new universe of vaccines suddenly becomes economically feasible to develop, and PopVax becomes the highest-value bet in global health R&D. Consider, for example, the case of the Hepatitis C Virus – HCV.
HCV & the case for PopVax as the highest-value bet in global health R&D
HCV is a blood-borne virus that infects the liver, often transmitted through needle sharing among injection drug users or improperly sterilized needles at medical facilities. It infects silently at first, and persists for years or decades before making its presence known in the form of cirrhosis, liver failure, and even liver cancer – 20% of liver cancer cases globally are caused by HCV.
The World Health Organization (WHO) estimates that there are 1.5 million new HCV infections every year across the world, resulting in 250,000+ deaths every year, with the burden concentrated in low- and middle-income countries. This death toll is despite the existence of curative antiviral treatments – since HCV screening is not routine, by the time symptoms appear, years of irreversible damage may have been done, and in many cases it may be too late to save the liver, and even the patient themselves.
Even when HCV is caught early, the antivirals are not a panacea. These 12-24 week regimens of up to 3 pills a day require rigorous adherence, a schedule that many of those most susceptible to the virus, including injection drug users, aren’t able to consistently maintain. In the US, HCV is particularly prevalent in the prison system thanks to needle sharing by drug users, unsafe tattooing practices, and frequent violence involving blood-to-blood contact, but the ~$50,000 cost of a full course of treatment means that prison systems ration care to only the sickest patients who already have advanced liver fibrosis. In lower-income countries, even the few hundred dollars the generic versions of these treatments cost may be prohibitive, and though governments have begun offering these medications for free, including in India, the lack of screening means that most infected individuals aren’t identified until late-stage disease, if ever. Compounding these problems, many of the highest-risk patients who are routinely exposed to the virus get repeatedly reinfected, and need to be screened and treated repeatedly.
An effective vaccine would be much, much cheaper to deploy – as little as $4/dose in lower-income countries, including the cost of administration – while saving hundreds of thousands of lives and preventing up to a million cases of irreversible liver damage each year. In the US, ~$5 billion is spent each year on HCV antivirals, so even at a cost of $50/vaccine dose administered, vaccinating 5 million of the highest-risk individuals each year, at a total cost of $250 million, would save over $2 billion each year if vaccination reduced the total case count by 50%.
Unfortunately, developing such a vaccine is anything but straightforward. Like HIV, HCV has an exceptionally high mutation rate, and exists as an ensemble of variants – a quasispecies – within a single individual. This genomic diversity allows the virus to continuously evade immune clearance, mutating around the antibody and T cell responses it elicits, and establish chronic infection that can be lifelong.
This biology makes HCV an unusually difficult vaccine target – any effective vaccine must not just protect against the particular strain of the virus that a person is initially infected with, which may hail from any of 7 distinct genotypes and many subtypes, but also confer broad protection against all possible variants the virus may mutate into in its quest to evade the immune response the body mounts against it. Most vaccines fail at the preclinical stage – since its discovery in 1989, 35+ years of research have yielded only 5 prophylactic HCV vaccine candidates that have made it to clinical trials, with only 1 ever progressing to an efficacy study, which failed.
There are many that despair of the possibility of ever developing a successful prophylaxis for this pathogen. They point to the tens of billions of dollars that have been spent on HIV vaccine development, where a similar problem of plenty in mutation and genomic diversity has defeated hundreds of distinct HIV vaccine candidates over the past 4 decades, as an ominous portent of what is to come. HCV, however, has an antigenic weakness that HIV doesn’t – the virion and its key entry proteins, E1 and E2, are fundamentally unstable, and many if not most mutations destabilize them to the point of nonfunctionality. As such, the space of mutations that we must combat with an HCV vaccine is much narrower than in HIV, since we can afford to ignore mutations that don’t preserve the pathogen’s ability to infect human cells. Further, unlike HIV, HCV doesn’t integrate into the host’s genome, which substantially increases the likelihood that the immune system can completely clear the infection.
These distinctions are borne out by observational data – there is strong evidence that over 20% of people who get infected are able to generate an immune response that spontaneously clears HCV, while only 0.5% of people infected with HIV are able to maintain undetectable viral levels without the use of antivirals, and even these so-called elite controllers of HIV almost always progress towards higher viral loads and serious symptoms of the disease. It’s much more likely that a vaccine can be developed to successfully replicate a response already elicited in 20%+ of people naturally than one found in only 0.5%.
At PopVax, we see HCV as a Goldilocks pathogen upon which to test and validate new immunogen design technologies – it’s difficult enough that legacy methods don’t work out of the box, but tractable enough that we should be able to detect some signal of progress.
This is a hard but solvable scientific problem attached to a vast and underserved public health need – precisely the sort of thing PopVax revels in attacking. We’ve applied all of our technology platforms and design methods, as well as our rapid wet lab data generation approach and machine learning feedback loop, to design and iterate on a series of HCV vaccine candidates based on a wide variety of scientific hypotheses. We have been relentlessly empirical in evaluating what works and what doesn’t, doubling down on the best-performing approaches, and after 3 cycles of testing, with our candidates improving at each iteration, we’ve achieved what we believe are among the best ever preclinical results in the field. In our pseudovirus neutralization assays, our v3 HCV RNA-encoded immunogen design elicited a neutralizing antibody titer more than 20 times higher than the reference soluble E2 immunogen, potently neutralizing at just a single dose, without any booster. Our goal is to be able to get a single candidate to elicit a neutralizing immune response against a diverse panel of pseudoviruses that is representative of the genomic diversity of the pathogen, with the hope that this breadth will be sufficient to confer protection in humans.
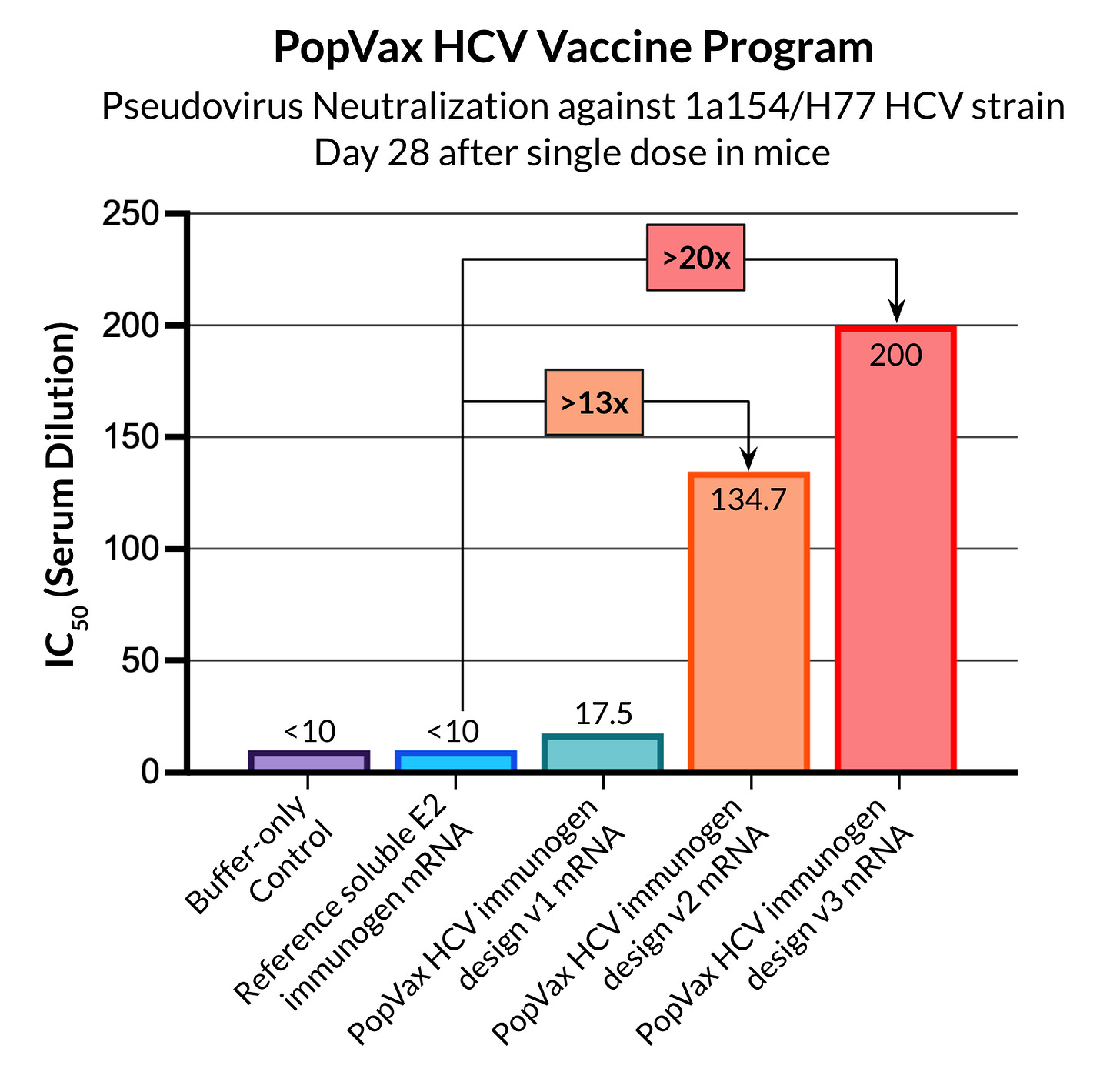
This is the absolute frontier of vaccine R&D, and we’re doing it all right here in Hyderabad. We are now iterating fast enough, and the results are improving quickly enough, that we believe we have a clear path to advancing an HCV vaccine candidate into a first-in-human trial by the end of this year or early next. The first candidate may not succeed at eliciting the immune response we are looking for, but it is cheap enough for us to keep putting these vaccines into clinic that we will keep at it until one does.
If we do succeed at developing an effective HCV vaccine, it will be a remarkably efficient mechanism to save lives. How efficient, exactly? This is the sort of question often contemplated by Givewell, an organization that exists because Peter Singer once asked a question so simple it was almost embarrassing – if you could save a drowning child’s life by jumping into a pond to rescue them at the cost of ruining your shoes and muddying your suit, are you not obligated to? The effective altruism movement that followed built an entire infrastructure for answering that question rigorously, ranking charities and publishing cost-per-life-saved estimates, bringing quantitative discipline to the otherwise chaotic business of doing good. The interventions they rank highest – mosquito nets to prevent malaria, vitamin A supplementation, deworming – can save a life for somewhere between $3,500 and $5,500, which is considered to be extraordinarily efficient. Vaccines are more efficient than these exemplars – GAVI’s pneumococcal vaccine Advance Market Commitment, a pioneering fund that successfully stimulated the development of affordable pneumococcal vaccines and oversaw the deployment of 1.7 billion doses of the vaccines across low- and middle-income countries, saving 600,000 lives, cost only $2500 for each life saved.
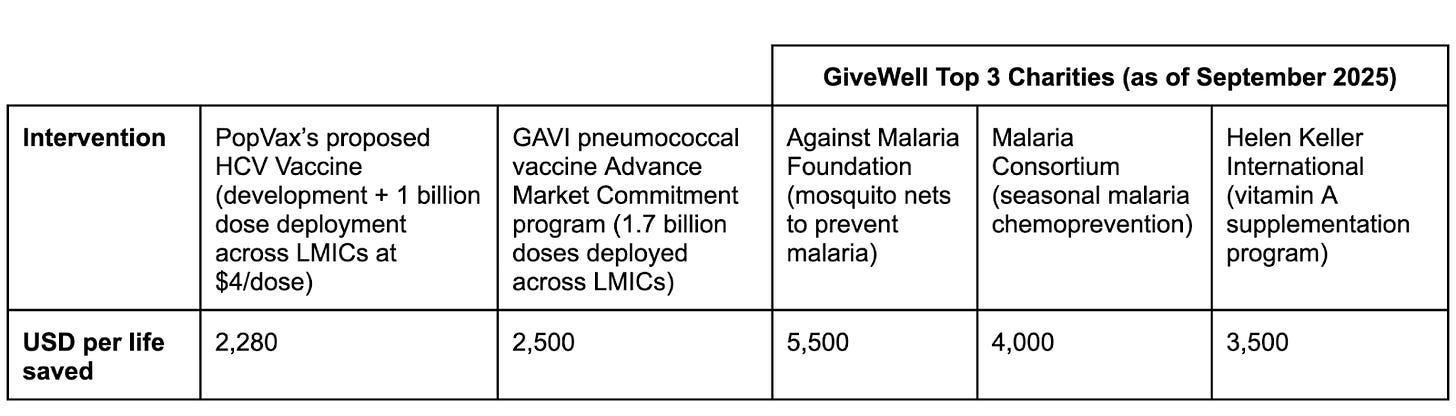
An effective PopVax HCV vaccine, developed and manufactured at Indian cost structures, would be even more efficient still – we could save a life for as little as $2280.
Here’s how: HCV, as mentioned above, kills ~250,000 people each year. Over 20 years, that totals 5 million lives lost, with lower-income countries like Bangladesh, China, India, Indonesia, Nigeria, and Vietnam collectively sharing a disproportionate burden of global deaths.
We would propose to deploy 1 billion doses of the vaccine in high-risk populations across these geographies over 20 years. If we assume that half the lives lost to HCV would be saved by that intervention, which is conservative given how concentrated HCV hotspots tend to be, we could save 2.5 million lives. We estimate that our proposed single-dose HCV vaccine could be priced at roughly $4/dose for lower-income countries, and we assume that the average deployment cost is $1.5/dose (going off numbers from the COVID-19 pandemic), yielding $5.5 billion of supply and deployment cost. We estimate that it would cost $200 million to develop the vaccine and bring it through clinical trials to approval in these key geographies. The total cost would then be $5.7 billion to save 2.5 million lives – $2280 per life saved.
The cost per life saved is, of course, dominated by the deployment cost, not the development cost, but the considerably lower development cost at PopVax is key to us being in a position to develop the vaccine at all. An HCV vaccine is unlikely to substantially exceed the $1 billion/year in sales needed to be considered a blockbuster, and our development cost of $200 million may not yield a successful vaccine – this is, of course, a very hard problem. Even if we estimate, however, that we only have a 20% chance of succeeding, and that the vaccine would only gross $500 million/year over 15 years, with future revenue beyond that uncertain due to patent timelines, we’d be investing $200 million upfront for an expected return of over $1 billion, assuming a reasonable profit margin, which is a bet we’re very willing to make.
Run this math from Boston or SF, and it breaks down entirely. Given the high cost of R&D in those geographies, you’re suddenly investing $1 billion in development costs for a probabilistic $1 billion return, an inconvenient reality that explains why there have been hundreds of HIV vaccine candidates in clinical trials, most from pharma companies and biotech startups, but only 2 prophylactic HCV vaccine candidates have ever been advanced to clinical trials by private companies.
What PopVax developing novel vaccines in India enables, then, is not simply a discount on the cost of doing science, but a fundamental change in what science is done at all – we are tearing down the financial wall that has kept an entire class of desperately-needed vaccine programs from being built at all. And that is just one program, one pathogen – the same logic applies to TB, to Strep A, and many other diseases that disproportionately afflict the poorest people on the planet. This math makes it possible for us to raise money to fulfil our Million Lives Mission without having to rely entirely on philanthropy, because our investors will make substantial returns if we succeed.
Of course, that same cold, hard financial math multiplies the public health ROI of funding PopVax philanthropically as well.
Once an effective vaccine is developed, governments, multilateral organizations, and philanthropies are extremely likely to find funding deployment to be a very compelling proposition, as they have done for relatively recently-developed vaccines such as HPV and malaria, but most of them are not willing to take the risk of funding early-stage development for a pathogen with HCV’s complexities.
That reticence provides a unique opportunity to funders willing to take scientific and technological risk – just $200 million of funding could tip the scales towards the development of an HCV vaccine that could save 2.5 million lives that otherwise might not exist. Taking into account our prior assumption of a 20% chance of success, that’s just $400 per life – the cost of a pair or two of Peter Singer’s shoes. If it cost $1 billion to develop the vaccine, as it might in the West, that would be $2000 per life just to get the ball rolling, at which point pneumococcal vaccines and Givewell’s less scientifically-risky recommendations might start looking much more attractive. The public health ROI of investing in PopVax’s vaccine R&D is utterly enormous, and the moment is perfectly poised for the right people to come along and unlock this door.
And, indeed, they have begun to. We’ve recently begun work on a new $3.5 million+ dollar research contract with the Gates Foundation to develop novel rabies and HPV vaccines, and a $1.5 million research contract from Renaissance Philanthropy to develop our HCV, TB, and Strep A vaccines. We’ve also raised $7.5 million+ in our first equity funding led by Facebook co-founder Dustin Moskovitz & Cari Tuna’s Good Ventures, advised by the good folks at Coefficient Giving (formerly known as Open Philanthropy), with personal investments from technology luminaries including Viswa Colluru, founder of the biotech unicorn Enveda, and Dhaval Shroff, who pioneered the pixels-to-actions AI architecture for Tesla’s self-driving cars. Our investors all believe that we have the potential to provide them substantial returns on their investment while saving millions of lives, and we don’t intend to let them down.
What’s next for PopVax? It’s time to accelerate.
Vaccine acceleration is needed now more than ever: we need step changes in vaccine technology to combat the increasingly difficult landscape of pathogens we face, both natural and engineered. Every year we allow things to proceed at their current glacial pace, millions of people die in expectation, and we leave open the possibility of truly catastrophic pandemic scenarios involving viral and bacterial respiratory pathogens that could make the COVID-19 era seem like a relaxed stroll in the park.
Unfortunately, US vaccine companies have been in the doldrums. Stock prices in the industry have fallen faster than birthrates in Europe, funding is harder to come by than it has been since the depths of the Great Financial Crisis of the late 2000s, and the new US regulatory regime has brought with it bold new ideas which, right or wrong, have created an uncertainty that threatens to upend the already cursed mathematics of vaccine development. The atmosphere in Boston & SF biotech was positively funereal last year, as mass layoffs rippled through big pharma and small startups alike.
PopVax is a glimpse at the future of vaccine development. This future will be unevenly distributed, as all futures are, but this time weighted towards India and the rising world, not away from us.
In the past, the best Indian biotechnologists were seldom plying their trade here in India. Top undergraduates and graduate students in biotech-related fields would exit the country to vie for academic and industry positions in the US, where they could get both the resources and the guidance they need to reach their full potential, but this door appears to be closing rapidly – not only is the US government looking to reduce immigration into the country across the board, with the widely-used H1B visa for skilled workers now requiring a $100,000 upfront payment to qualify for, but it is also increasing scrutiny of and cancelling visas for PhD students, many of them Indian. At the same time, the National Science Foundation (NSF) and the National Institutes of Health (NIH), which fund much of the early-stage bio research in the US, have frozen billions of dollars of grants in order to substantially shift their priorities, compromising the ability of many leading academic labs to retain their students and postdocs, and making it difficult for them to operate at the same scale and speed as before.
As one of the few companies developing truly novel biotechnology in India, we are now inundated with emails and LinkedIn messages from people who suddenly find themselves in an uncertain position in the US, and are now trying to come to grips with whether and how they might operate in India. In particular, they are concerned that there are few opportunities for them to continue to do novel work in India – that if they return, they’ll be stuck in the same generics-biosimilars-CRO-CDMO grind that they left to escape from in the first place.
Fortunately, PopVax can promise them the chance to work on truly novel R&D problems at the frontier of what is possible – beyond our own ideas, we also see an opportunity to collaborate with some of the best vaccine researchers in the US to bring their best ideas to life rapidly here in India, for the mutual benefit of both countries, including fulfilling the geopolitical imperative of ensuring that the next generation of vaccines don’t exclusively come from China. We’ve now assembled a pipeline of returning talent who have given us a delightful shot in the arm – pun very much intended – enabling and motivating us to move faster than we’ve ever moved before to realize the Million Lives Mission. The time to v/acc has come!
PopVax’s Phase I clinical trial of PVX-001 in Australia in a few months will provide powerful validation of our platform and design approach, and will serve as a springboard from which we plan to launch not one, not two, not three, but six more vaccine programs into the sky, including HCV, TB, Strep A, and malaria. We intend to use innovative regulatory frameworks such as those established in Próspera, the charter city and special economic zone in Honduras, to initiate small-scale first-in-human trials of all six of these new vaccine programs by the end of this year or early next while upholding the highest standards of participant safety. If we succeed, this will likely be the fastest any biotech startup has ever translated this scale of novel vaccine candidates into clinical trials.
The same technologies we are applying to these challenging diseases may well be the key to helping us combat the threat of engineered pathogens, allowing us to build a library of vaccines and other anti-infectives that are already able to combat all relevant families of viral and bacterial pathogens from which potential bioterrorists and the generative models they use draw their inspiration, which can then be continuously updated and improved in response to the expanding biological design capabilities of frontier AI models. An effective defense requires a full-stack approach of the kind that PopVax is built to provide: zero-shot AI-powered design coupled with rapid validation and GMP production in an adaptable, always-on foundry.
The opportunity in front of us is not modest, and neither is the weight of the moral responsibility on our shoulders. Every year that passes without vaccines against HCV, TB, and Strep A, every year that malaria vaccines are not as good as they could be, millions of people die who did not have to. Every year we fail to build the rapid-response infrastructure that a future engineered pandemic will demand, we leave humanity’s very future hanging in the balance. The tools to change this – generative AI for biological design, programmable RNA, precision delivery, human immune organoids – exist right now, in our hands, right here in Hyderabad, at the beating heart of the rising world.
Over the next decade, PopVax aims to fulfill the Million Lives Mission by developing vaccines that save one million lives each year. Within the next 50 years, we aim to do nothing less than build a bridge over the troubled waters of our stormy present into our glorious future in which no man, woman, or child need fear infectious diseases.
Into that heaven of freedom from pathogens, let humanity awake.
Come help us save one million lives each year and build the foundations of a pathogen-free future – apply at popvax.com/jobs, or email work [at] popvax [dot] com.
I’m Soham Sankaran, the founder & CEO of PopVax. Feel free to email soham [at ] popvax [dot] com if you’re interested in discussing vaccine acceleration. I was also recently appointed a fellow at Renaissance Philanthropy to explore setting up a Vaccine Renaissance Fund to accelerate the translation of frontier vaccine technologies into the clinic – feel free to reach out if you’re a vaccine researcher or funder interested in collaborating on this.
Dhruv Arora, a program manager at PopVax, made substantial contributions to the writing of this piece.
You can also follow PopVax on Twitter/X, Bluesky, LinkedIn, and YouTube, or email us at hello [at] popvax [dot] com.


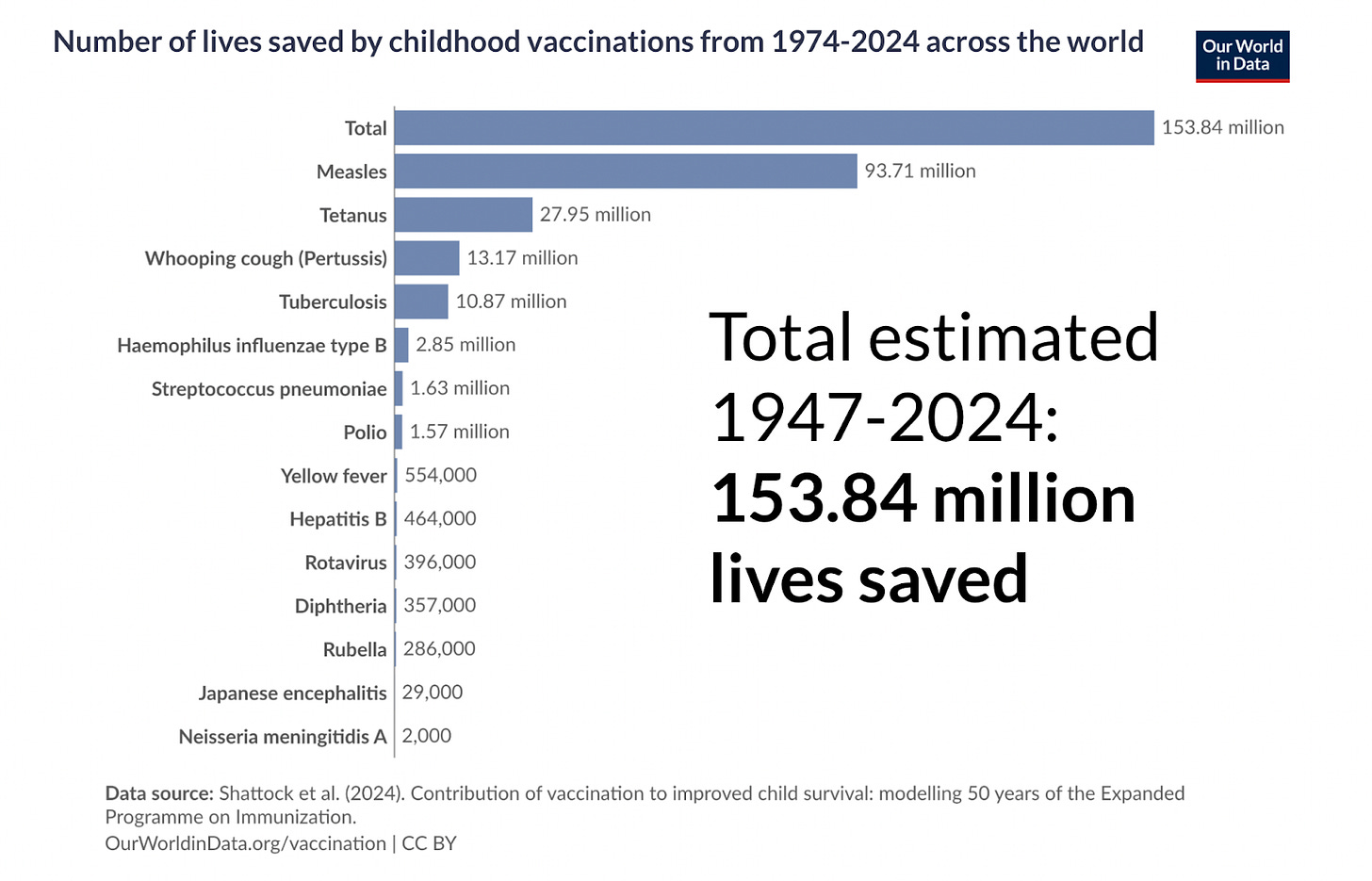




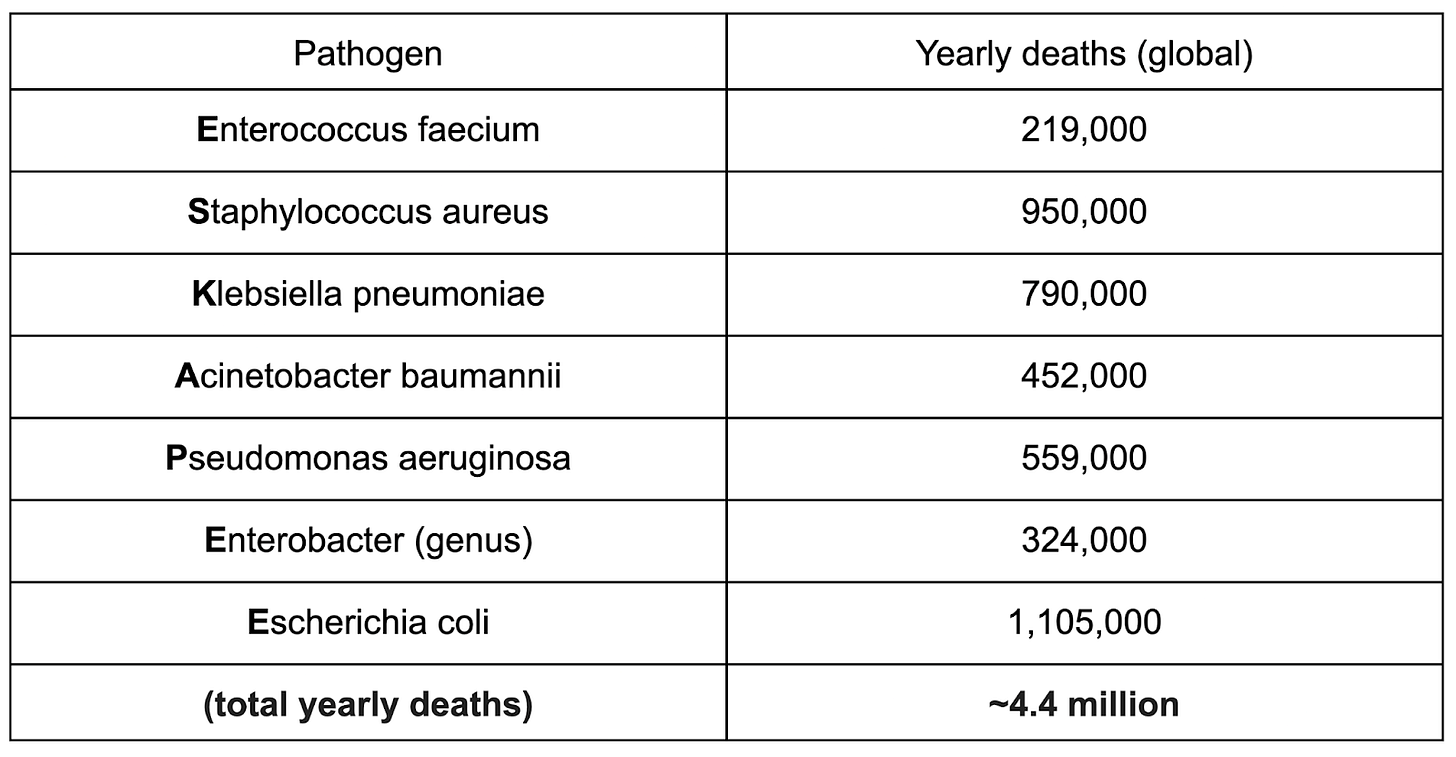
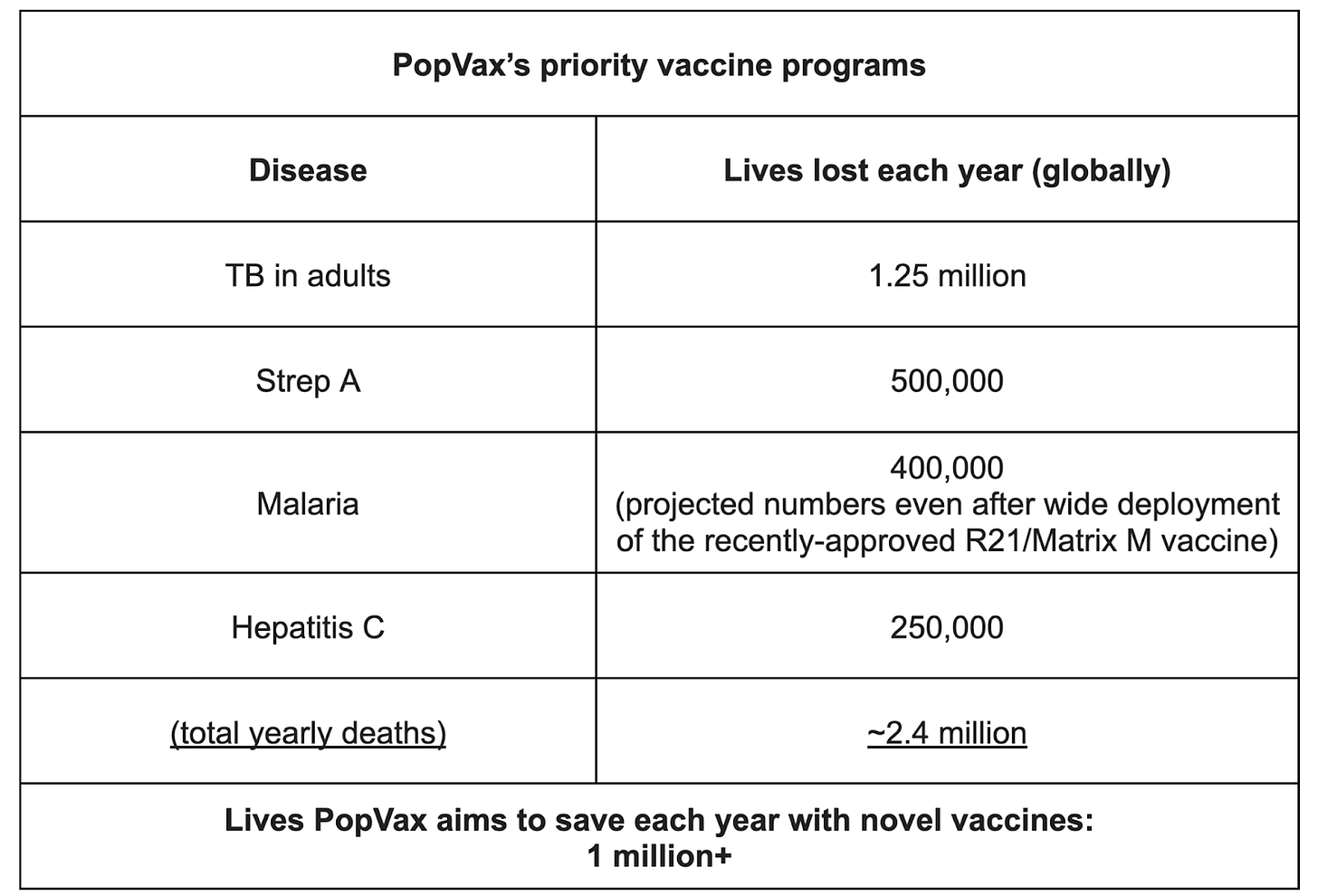


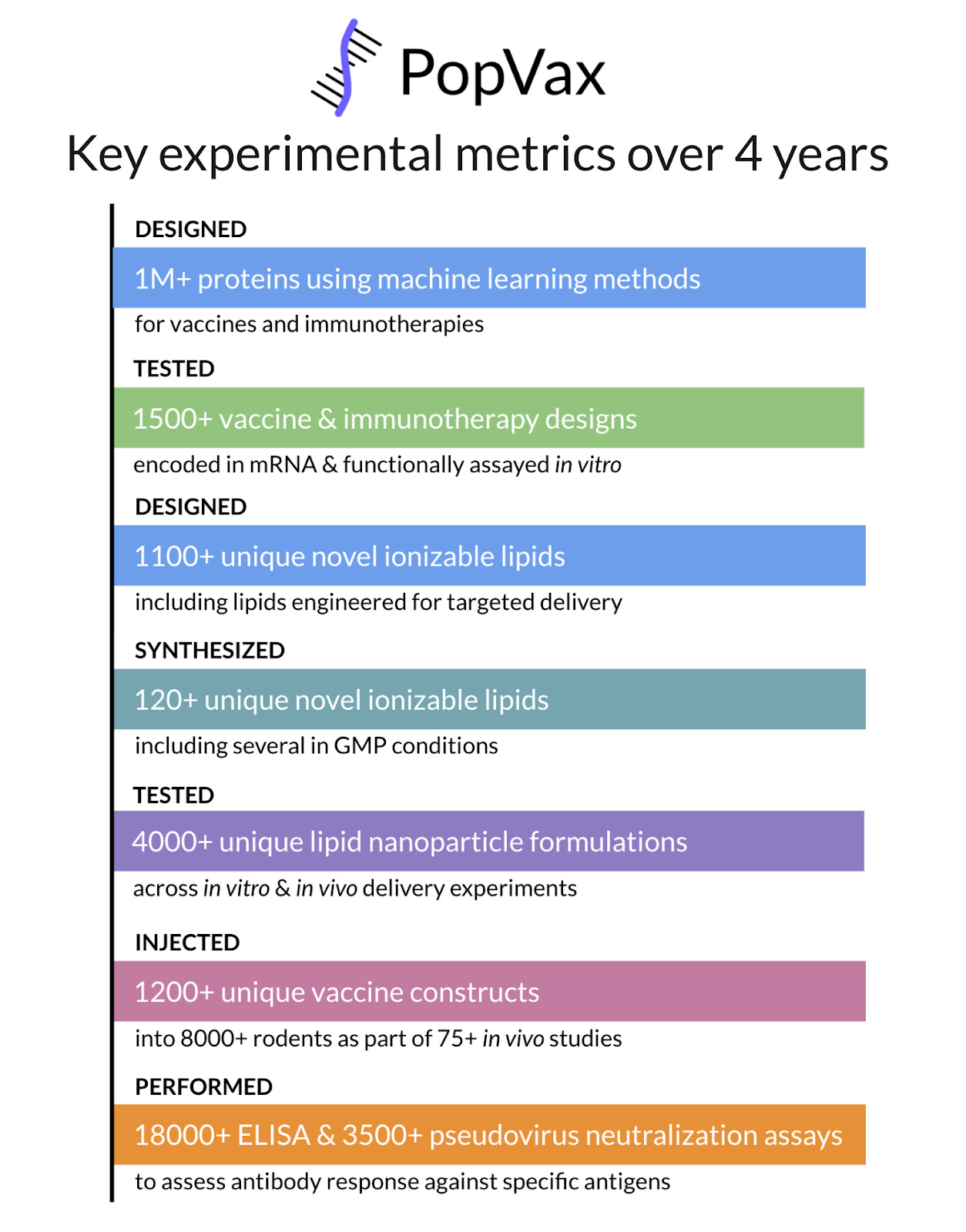
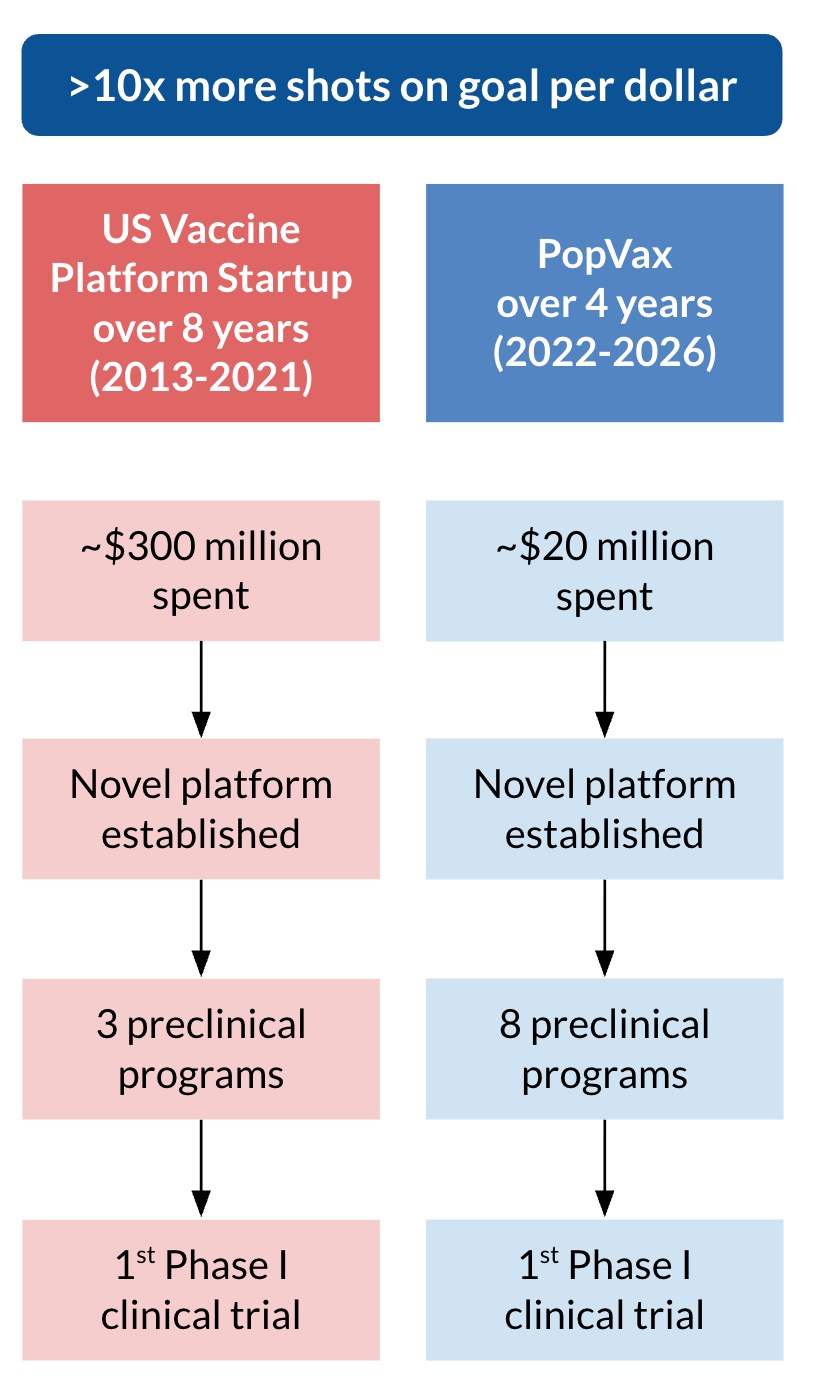
Great article! Learned a lot. Good luck! What a clear and compelling vision!
Alphafold2 is gem.